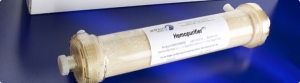
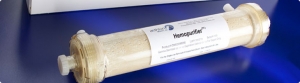
The device from San Diego-based Aethlon Medical Inc. targets the rapid elimination of the virus and immunosuppressive proteins from the circulatory system of infected individuals.
Aethlon has shipped four Hemopurifiers to the Biocontainment Unit at Nebraska Medical Center (NMC) in Omaha as a therapeutic option for Ebola patients that may be treated there. The company has provided onsite training to NMC clinical personnel related to the implementation of Hemopurifier therapy within the dialysis infrastructure already located at the center.
The Nebraska Biocontainment Unit was commissioned by The United States Centers for Disease Control (CDC) in 2005. It is a joint project involving Nebraska Medicine, Nebraska Health and Human Services, and the University of Nebraska Medical Center. It was designed to provide the first line of treatment for people affected by bioterrorism or extremely infectious naturally occurring diseases such as Ebola. It's the largest facility of its kind in the United States. The NMC has treated three Ebola patients since the current outbreak.
Aethlon's Hemopurifier therapy previously was used on a Ugandan doctor infected with Ebola virus and treated at the Frankfurt University Hospital in Germany. At the time Hemopurifier therapy was administered, the doctor was unconscious and was suffering from multiple organ failure. His viral load prior to the administration of a single 6.5-hour Hemopurifier treatment was measured at 400,000 virus copies per milliliter of blood (copies/ml). Post-treatment viral load was measured at 1,000 copies/ml and never again rose above that level, according to the company. Viral load became undetectable five days after therapy. The treatment was well tolerated with no adverse events reported. Additionally, a post-treatment elution protocol verified that 242 million Ebola viruses were captured within the Hemopurifier during treatment. The doctor has since made a full recovery and has returned home to his family in Uganda.
In the United States, Hemopurifier therapy is available to treat Ebola patients through U.S. Food and Drug Administration-expanded access "emergency use" provisions to address life-threatening circumstances for which an alternative therapy is not available. At present, no antiviral therapy or vaccine has proven to be effective against Ebola virus infection in humans. Aethlon is providing Hemopurifiers to the NMC on a humanitarian basis and therefore will not generate revenues as a result of its initial shipment.
Aethlon soon will begin the first U.S. clinical Hemopurifier studies following the FDAs approval of an Investigational Device Exemption (IDE) application. The study will contribute safety data to advance the device as a broad-spectrum countermeasure against bioterror and pandemic threats, including Ebola and chronic viral pathogens such as HIV and Hepatitis C (HCV).
Aethlon Medical creates targeted therapeutic devices to address infectious disease, cancer and neurodegenerative disorders. The company's lead product is the Aethlon Hemopurifier, a first-in-class device that selectively targets the rapid elimination of circulating viruses and tumor-secreted exosomes that promote cancer progression.