07.18.13
San Diego, Calif.-based Aethlon Medical Inc., a company that develops selective therapeutic filtration devices to address infectious disease, cancer and other life-threatening conditions, has appointed Chetan S. Shah, M.D., to its board of directors.
Shah is a board certified otolaryngologist (an ear, nose and throat specialist). He is an advisory board member at The Bank of Princeton, and a founder, partner and board member of the Surgery Center at Hamilton, Physician Management Systems and Princeton Eye & Ear, all in New Jersey. Shah also serves on the board of two other private companies. He holds teaching positions and serves on multiple hospital committees in the area and is on the Audiology and Speech Language Pathology Committee for the state of New Jersey. Shah received his bachelor’s degree and medical degree from Rutgers University and Robert Wood Johnson Medical School, both in New Brunswick, N.J.
“We are honored to have Dr. Shah join our board,” said Jim Joyce, chairman of Aethlon. “His wealth of medical and business experience will be instrumental in guiding the future growth and achievement of our organization.”
“Aethlon Medical’s Hemopurifier is first in class therapeutic device that is a game changer in both the infectious as well as cancer treatment regimen,” Shah said. “As a physician, I am excited at the prospect of being able to help millions of patients worldwide. As a director, I look forward to building value for all who have patiently supported the company over the past decade.”
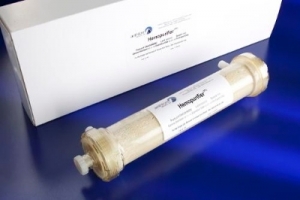
The Aethlon Hemopurifier, a blood-filtering device, has broad-spectrum capabilities against viral pathogens, including the human immunodeficiency virus (HIV), hepatitis C virus (HCV) and numerous bioterror and pandemic threats, according to the company. Human studies have demonstrated the Hemopurifier to be safe and provide average viral load reductions of greater than 50 percent during four-hour treatment periods in both HCV and HIV infected individuals without the administration of antiviral drugs. The device is currently the subject of a human clinical study in India to evaluate its ability to accelerate viral load depletion when used in combination with HCV standard of care drug therapy. An investigational device exemption to initiate clinical studies in the United States is pending with the U.S. Food and Drug Administration.
Shah is a board certified otolaryngologist (an ear, nose and throat specialist). He is an advisory board member at The Bank of Princeton, and a founder, partner and board member of the Surgery Center at Hamilton, Physician Management Systems and Princeton Eye & Ear, all in New Jersey. Shah also serves on the board of two other private companies. He holds teaching positions and serves on multiple hospital committees in the area and is on the Audiology and Speech Language Pathology Committee for the state of New Jersey. Shah received his bachelor’s degree and medical degree from Rutgers University and Robert Wood Johnson Medical School, both in New Brunswick, N.J.
“We are honored to have Dr. Shah join our board,” said Jim Joyce, chairman of Aethlon. “His wealth of medical and business experience will be instrumental in guiding the future growth and achievement of our organization.”
“Aethlon Medical’s Hemopurifier is first in class therapeutic device that is a game changer in both the infectious as well as cancer treatment regimen,” Shah said. “As a physician, I am excited at the prospect of being able to help millions of patients worldwide. As a director, I look forward to building value for all who have patiently supported the company over the past decade.”
The Aethlon Hemopurifier, a blood-filtering device, has broad-spectrum capabilities against viral pathogens, including the human immunodeficiency virus (HIV), hepatitis C virus (HCV) and numerous bioterror and pandemic threats, according to the company. Human studies have demonstrated the Hemopurifier to be safe and provide average viral load reductions of greater than 50 percent during four-hour treatment periods in both HCV and HIV infected individuals without the administration of antiviral drugs. The device is currently the subject of a human clinical study in India to evaluate its ability to accelerate viral load depletion when used in combination with HCV standard of care drug therapy. An investigational device exemption to initiate clinical studies in the United States is pending with the U.S. Food and Drug Administration.