Mark Crawford02.01.10
An Evolving Role
Is machining in medical device manufacturing an advanced science or a dying art?
Mark Crawford
It’s a demanding industry out there. That’s no secret. OEMs want products and parts to be better, faster and cheaper—all the time. They want to drive down costs without reducing functionality. Manufacturers and suppliers continuously are challenged by more complex designs, combined materials and miniaturization, all of which present different heating, temperature and sterilization issues. More designers expect good advice or suggestions from key members in their supply chain about the manufacturability of their products. Further, designers are expecting prototype parts to be delivered as quickly as one or two weeks from notification.
“To go from the designer’s solid model to finished part means an increased level of expertise, right down to the shop floor,” observed Al LaVezzi, owner of LaVezzi Precision in Glendale Heights, Ill.
According to LaVezzi, the trend toward laparoscopic and other minimally invasive surgical techniques (i.e., smaller, lighter weight and more complex devices) has required his company to increase its capability to machine smaller, tighter-tolerance parts, which means purchasing more sophisticated machining equipment.
Trish Mowry, CEO of Metal Craft and Riverside Machining and Engineering in Chippewa Falls, Wis., agreed.
“The biggest challenge today is simply finding ways to meet the increasing requirements of our customers—cost reduction, more documentation and disclosure, shorter manufacturing times and more complex products,” Mowry said. “Tighter controls are being placed on the contract manufacturer, locking processes to single machines, which take away our ability to be flexible in manufacturing and to keep costs down. This also hinders our ability to shorten the manufacturing window to allow our customers to be the first to market. And of course there is the cost reduction that is also desired. While there may be options that new equipment or processes may bring, that is not realistic for most manufacturers with the current economy.”
|
Because of increasing demands for advanced materials and composites, George Weaver, vice president of marketing for Precision Medical Products in Denver, Pa., sees a trend away from total machining of medical devices.
“There is a great deal of economic pressure on medical device manufacturers to reduce costs,” he said. “Parts made from combined materials are starting to replace totally machined parts. There is a trend toward using lower-cost, injection-molded plastic disposables over machined reusables, for example. As an OEM medical manufacturer, we are receiving lots of inquiries for composite devices that combine metal and plastic. This is actually giving us more capacity—we’re not machining as much, but overall our business is increasing.”
Leading Technologies
Products are being designed to take advantage of full five-axis machining technologies, which means strong programming, process and material knowledge are vital for contract manufacturers.
The advantages of having a multi-axis machine are not maximized unless they are operated by creative programmers who can get the most out of the machine and challenge what it can do.
“Having a five-axis machine does not mean a lot if you don’t have the skilledpersonnel to program and develop the machine/process to produce volume consistently,” Schafer pointed out. “We have seen companies being limited by the machine platform—cheaper platform—and the skill level of their programmers. Operating five-axis linear machines—no ball-screw movements—equates to greater machine accuracy, repeatability and surface finish on the materials being used in this market. We use Siemens PLM for most of our machine programming—a very challenging software that requires a high skill level. Many companies use ‘canned’ programming platforms that can also limit what can be done on a part that combines complex geometry and close tolerances.”
Multi-axis machining has been proven to significantly reduce the setup times required, compared to producing the same part via multiple machining operations. “For example,” noted Jeff Shepherd, director of operations and engineering for Sandvik Medical Solutions in Memphis, Tenn., “we were able to eliminate three set-ups recently on a customer’s product, which resulted in significant savings and lead-time improvement.”
Some companies are making more use of water-jet and laser cutting to obtain nearer to net shaped blanks.
“Water-jet machining is not new, but it has a proven cost savings relative to milling, for the cutting of shapes in tough materials,” said David Cabral, president of Five Star Manufacturing in New Bedford, Mass.
Metal injection molding (MIM) is another existing technology that is “starting to catch on now,” Weaver added. “Created in the 1970s, MIM is a process whereby a slurry of metal powders and plastic binders is injected into a mold using injection-molding equipment. The resulting ‘green’ solid is then put in an oven, which drives off the plastic and leaves the metal part.” The final product is typically 95 to 98 percent the density of wrought metal and has similar mechanical properties and corrosion resistance. MIM also is compatible with many ferrous, nickel-based and copper-based alloys.
“The big advantage is time,” Weaver said. “After the mold is made, you can pump out a metal part about every minute. New resins are also being developed that have better properties and improve the overall process.”
Precision Medical Products also recently added laser technology to its arsenal of production weapons for welding together metal components. “In the past, we might have tried to manufacture a complicated part by welding the pieces together—today we can use the laser,” Weaver said. “We can also use laser technology to engrave identification or lot numbers on implants and other parts.”
A five-axis laser welding machine costs about $200,000 and a laser-engraving machine about $100,000.
Advanced Materials
Products can be a challenge to machine for a number of reasons, including the material type, finish requirements, dimensional tolerances and assembly. “For orthopedics,” Shepherd said, “this is limited to minimally invasive surgical approaches that require more nimble instrumentation. In some cases it has resulted in a tightening of tolerances due to the requirements of the instrument to produce the result via keyhole-type applications.”
For LaVezzi, the most challenging components are those that must be manufactured from high-nickel-bearing alloys that have “small, tightly toleranced details and holes,” he said.”The spindle quality—rigidity and concentricity—and the tool quality are extremely important in these cases. Thankfully, there are some really great manufacturers of tools that are constantly increasing their selections and grades.”
Cabral reported that Five Star Manufacturing is seeing a significant increase in the use of carbon fiber in the manufacture of devices because of its high-strength, low-weight and radiolucent (near invisibility to X-ray) properties. The use of carbon fiber allows a surgeon to shoot an X-ray while the device is in place, eliminating the need to break down the instrument setup to check the alignment of an implant or some other component.
“Carbon fiber is a challenge to machine because of its inherent properties,” Cabral explained. “It is a very abrasive material to machine, and diamond tools need to be used to maximize manufacturing efficiency. We have tried other lower-cost cutting tools, but diamond has proven to be the best for wear and cost effectiveness.” The use of advanced materials often increases the complexity of the manufacturing process and sometimes requires new cutter technologies.
“The precision of the devices warrants CMM [coordinate measurement machine] measurement versus conventional inspection procedures and the types of machinery used have moved to a higher level now, requiring surfacing, five-axis capabilities and other state-of-the-art techniques,” continued Cabral. “Carbon fiber is as strong as stainless steels but lighter in weight. It does not require new machinery, but is best machined using advanced cutter technologies.”
Products that are designed using advanced memory or wear materials such as nitinol, cobalt/chrome and titanium/carbide on bearing surfaces also are challenging to work with.
“Machining these materials effectively requires in part a reliance on our ‘tribal knowledge,’ which has been accumulated over 60 years of working with hard materials such as tungsten carbide and ceramics,” Schafer said. “It is difficult to replace experience and the tricks of the trade that come along with that cannot be underestimated. That said, we also incorporate new technologies such as our own advanced manufacturing process called molecular decomposition process (MDP).”
Schafer explained that MDP is a non-contact machining technology that provides the ability to achieve micron-level manufacturing tolerances. It allows the manufacturer to produce superior surface finishes. It also has a unique ability to not generate heat into the work piece, which makes it especially compatible with heat-sensitive materials such as nitinol.
“We have incorporated this technology with great success in machining these materials for recent product launches,” he added. “MDP has been commercialized and is being used in the production of volume implants and instruments with very consistent, predictable and repeatable results. The fact that MDP is a non-contact process that does not create heat during manufacturing makes it an effective solution for difficult-to-machine materials like nitinol, titanium, carbide, cobalt/chromium and stainless.”
Regulatory Demands
Healthcare reform and the discussion surrounding comparative effectiveness will continue to put pressure on OEMs and challenge contract manufacturers to provide high-quality, cost-effective products that are safe for patients. In general, contract manufacturers are not as scrutinized by federal agencies as the companies that actually control the designs. However, as contract manufacturers get more involved in the design and process development phases, they will be scrutinized more closely to ensure compliance with proper procedures, documentation and validations to guarantee patient safety and minimize potential for litigation.
“Five Star responds by continually reviewing our internal quality systems and ensuring we close any gaps which can be left to failure of the system, resulting in a product quality issue,” Cabral said. “We have a dedicated quality group that provides the necessary resources to maintain the required standards.”
Contract manufacturers also reported that their clients are asking for more robust validation efforts, especially for Class III products.
“OEMs have become much more sensitive to manufacturing process changes for Class III as a result of increased pressure from the FDA,” Shepherd said. “As a result, once a process is locked down for a Class III device, it is very difficult to change the process, even if the change produces a better and/or less expensive part.”
In particular, Shepherd noted that customers want to do business with suppliers who exhibit the ability and dedication for tight regulatory compliance, including validation efforts.
“A supplier's ability to demonstrate effective management of a validation master plan for its respective facility is one such example,” he said. “This means that once the process is defined and parts are produced via that process, a manufacturer must justify any process changes which could result in having to resubmit to the FDA.”
Many contract manufacturers expect to see an increased effort by OEMs to audit their process controls and validation activities, as well as insist their contract manufacturers can demonstrate their understanding and investment in these systems.
“We think they will hold contract manufacturers to a much higher standard,” Schafer said. “We have been told that OEMs want to be able to ‘fly their company flag’ alongside the flag of their contract manufacturers. That means from a quality systems perspective you’d better have your act together and use this same mentality with any of your sub-suppliers, including auditing them and understanding what it means to have a validated manufacturing operation.”
OEMs want to maximize process management and reduce risk to ensure the most consistent flow of safe products. Instruments are now undergoing the same level of scrutiny that implants do. “There is no distinction between the two anymore,” added Schafer. “In order to succeed long term, you need to have a strong quality management system in place and very capable people, processes and machine technologies. The barriers to entry are very high for companies who currently work outside of this space—having the right equipment to make medical devices is only part of the total package.”
Keeping up with proper certification is a must for securing and maintaining business. “We are already certified to ISO 13485:2003 and 9001:2008,” LaVezzi said. “Some customers, however, are taking a lead from the automotive industry and requiring production part approval process, including part submission warrants to ensure a new product meets spec right out of the box. This ultimately ensures a smooth transition from prototype to production, which is good for both the customer and the company.”
Keeping Costs Down
Competitive pricing has always been a key driver for OEMs. “I think OEMs will focus even more on maintaining their margins in the years ahead and will look to leverage contract manufacturer pricing to aid them in their margin growth and preservation initiatives,” predicted Schafer.
Customers are involving suppliers more in the early design stages to gain their design for
Customers are involving suppliers more in the early design stages to gain their design for
|
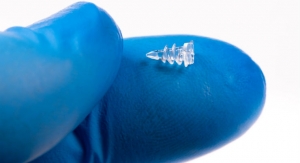
Threaded nitinol trocar guidewire produced from .040 inches diameter nitinol wire with a pitch of 101 threads per inch. The product shown was produced in a single pass grind without secondary burr removal or required electro-polishing operations. Photo courtesy of Oberg Industries. |
manufacturability input, “which reduces costs and lead times,” Shepherd said. “Most of the larger OEMs manage their own design function. However, suppliers with compatible CAD systems can work with these OEMs to suggest design changes for manufacturability and cost.”
Contract manufacturers also can help OEMs take advantage of lower-cost stam-ped components, which are then five-axis-machined to add unique features that cannot be stamped. This type of hybrid manufacturing solution can minimize costs and still provide designers with unique product advantages.
“There are certain applications where companies can save 40 to 50 percent with a stamped/machined part as compared to a 100 percent machined component,” Schafer said. “The challenge is understanding the limitations of progressive stamping and getting OEMs comfortable with designing products that can take advantage of the production speed and cost advantages of stamped components.”
Contract manufacturers also can help OEMs take advantage of lower-cost stam-ped components, which are then five-axis-machined to add unique features that cannot be stamped. This type of hybrid manufacturing solution can minimize costs and still provide designers with unique product advantages.
“There are certain applications where companies can save 40 to 50 percent with a stamped/machined part as compared to a 100 percent machined component,” Schafer said. “The challenge is understanding the limitations of progressive stamping and getting OEMs comfortable with designing products that can take advantage of the production speed and cost advantages of stamped components.”
|
|
To control costs, Sandvik Medical Solutions is working on two fronts: new product development and introduction and continuous improvement (CI) efforts for existing products. Sandvik utilizes program managers to liaise between customers, sales, engineering, quality, operations and supply chain to improve the overall management of the new product development and introduction process.
“We do not design any medical devices, but we do develop new materials for medical and industrial applications,” Shepherd said. “Our CI efforts are highly focused on lean manufacturing initiatives in all of our facilities. We are currently pursuing cellular manufacturing in an aggressive manner with a focus on speed, which will result in lower waste and significantly improved lead times.”
Sandvik Medical Solutions recently implemented an IT-driven “one plant” initiative that integrates the enterprise resource planning systems between its facilities. The first phase of this initiative will integrate its Tennessee and Alabama operations and result in significant reductions in administrative and supply chain costs.
Outsourcing to low-cost countries will continue to be a key strategy for some OEMs in the medical technologies market. Several large OEMs already have established manufacturing centers in these countries, primarily as a way to service local markets. “As experience is gained and their comfort level increases with their own and other offshore contract manufacturers in terms of quality, speed and pricing, we believe the outsourcing trend will continue in a controlled manner,” Schafer said.
“We do not design any medical devices, but we do develop new materials for medical and industrial applications,” Shepherd said. “Our CI efforts are highly focused on lean manufacturing initiatives in all of our facilities. We are currently pursuing cellular manufacturing in an aggressive manner with a focus on speed, which will result in lower waste and significantly improved lead times.”
Sandvik Medical Solutions recently implemented an IT-driven “one plant” initiative that integrates the enterprise resource planning systems between its facilities. The first phase of this initiative will integrate its Tennessee and Alabama operations and result in significant reductions in administrative and supply chain costs.
Outsourcing to low-cost countries will continue to be a key strategy for some OEMs in the medical technologies market. Several large OEMs already have established manufacturing centers in these countries, primarily as a way to service local markets. “As experience is gained and their comfort level increases with their own and other offshore contract manufacturers in terms of quality, speed and pricing, we believe the outsourcing trend will continue in a controlled manner,” Schafer said.
With the down economy, other OEMs are being more cautious, especially those that possess internal manufacturing capabilities.
“They are reducing outsourcing and choosing instead to better utilize their own capacities,” according to Shepherd. “OEMs who do not possess internal manufacturing capabilities do not appear to be focused on moving into manufacturing. They are instead focusing on their core competencies of product development, sales and marketing of medical devices.”
According to Cabral: “I know of some companies that are more design services suppliers who will look at a company like Five Star to provide machining services. We, as a contract machining supplier, do not commonly outsource machining or other processes that we would rather control in-house. Some processes, due to the high capital involved or presence of hazardous materials, are outsourced regularly to facilities that have the proper technologies and environmental controls.”
In an ironic twist, machining opportunities are actually on the rise in the United States because of the eroding value of the U.S. dollar. A number of overseas machining operations are now looking at coming to the United States because costs will be less.
“Right now we are working on three possible projects with manufacturers who had been offshore in Costa Rica, Austria and Germany,” Weaver said. “They want to do business in the U.S. because of the favorable exchange rate with the U.S. dollar."
A commitment to continuous improvement is essential to surviving (or thriving) in this brutal economic climate. Maintaining an approved supplier status is highly important, not only for the supply chain benefits but also to stay in business and fend off the competition.
“Not only must costs be managed relative to fixed expenses, but we must also track our production, outside labor and material costs as well, because these are what drive up our customer prices,” Cabral said. “Our customers are always looking to reduce their costs and expect us to assist them in this endeavor. We are being asked to look at everything we do. Only through continual focus and improvement in how we manufacture can we squeeze the maximum margins from the process.”
“They are reducing outsourcing and choosing instead to better utilize their own capacities,” according to Shepherd. “OEMs who do not possess internal manufacturing capabilities do not appear to be focused on moving into manufacturing. They are instead focusing on their core competencies of product development, sales and marketing of medical devices.”
According to Cabral: “I know of some companies that are more design services suppliers who will look at a company like Five Star to provide machining services. We, as a contract machining supplier, do not commonly outsource machining or other processes that we would rather control in-house. Some processes, due to the high capital involved or presence of hazardous materials, are outsourced regularly to facilities that have the proper technologies and environmental controls.”
In an ironic twist, machining opportunities are actually on the rise in the United States because of the eroding value of the U.S. dollar. A number of overseas machining operations are now looking at coming to the United States because costs will be less.
“Right now we are working on three possible projects with manufacturers who had been offshore in Costa Rica, Austria and Germany,” Weaver said. “They want to do business in the U.S. because of the favorable exchange rate with the U.S. dollar."
A commitment to continuous improvement is essential to surviving (or thriving) in this brutal economic climate. Maintaining an approved supplier status is highly important, not only for the supply chain benefits but also to stay in business and fend off the competition.
“Not only must costs be managed relative to fixed expenses, but we must also track our production, outside labor and material costs as well, because these are what drive up our customer prices,” Cabral said. “Our customers are always looking to reduce their costs and expect us to assist them in this endeavor. We are being asked to look at everything we do. Only through continual focus and improvement in how we manufacture can we squeeze the maximum margins from the process.”
Fortunately, material costs have decreased and been relatively stable over the past year, making pricing and budgeting easier. Cutting out waste by applying lean manufacturing principles and other quality-control measures is highly effective in reducing production costs and increasing efficiency and throughput. Any step or process that does not add value for the client should be fixed or eliminated.
“These initiatives are absolutely essential for eliminating waste in the process and being able to catch quality issues at the onset,” Cabral stressed. “Add in the benefit of faster turnaround and on-time delivery, and you have a recipe for long-term success. If companies are not looking for new methods to produce quality products more efficiently, they’d better get moving. Our domestic competitors are doing it, and our overseas competitors have used these initiatives for years. If you are not moving forward, you are falling behind.”