Sam Brusco, Associate Editor04.22.24
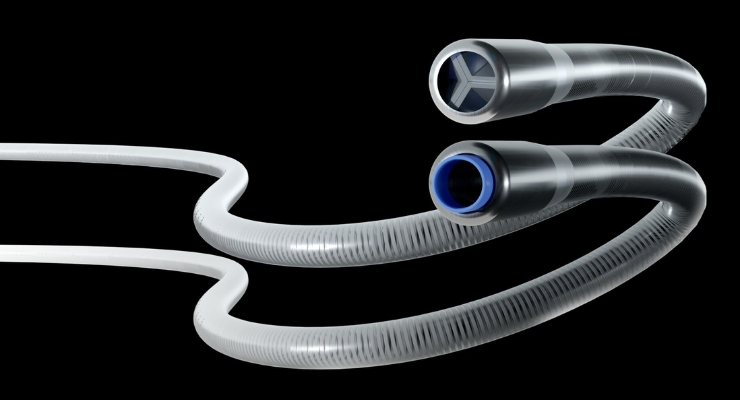
Expanse ICE has earned 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its ICE aspiration system, bringing a new player into the peripheral thrombectomy market.
The ICE system, according to the company, addresses complex challenges related to peripheral thrombectomies. Eitan Konstantino, Ph.D., founder and CEO of Expanse Medical, said the ICE catheter system was engineered to harness the aspiration power found in a large bore catheter, within the slender profile of a much smaller device.
"It is clear this device was built with physicians in mind,” Dr. Michael Lichtenberg, chief medical officer of angiology at the Vascular Center Clinic in Arnsberg, Germany, told the press. “It aims to address some of the biggest issues we see regularly. The device was designed and built with a strong understanding of foundational physics that leaves me optimistic for its success.”
Expanse ICE is a company born from Expanse Medical, a medical device incubator founded by Dr. Konstantino. The team that built the Expanse ICE system also built AngioSculpt, Chocolate, and other neurovascular products. The company focuses on developing solutions for vascular diseases.
"I am looking forward to being among the first users of the Expanse ICE system. Its ability to offer powerful clot removal in a compact size is an intriguing proposition that could significantly enhance our ability to meet patient needs more effectively and improve treatment outcomes in peripheral vascular disease," said Dr. Aravinda Nanjundappa, director of peripheral vascular Interventions at the Cleveland Clinic.
"The thrombectomy market is witnessing robust growth and the approval of the ICE system comes at an auspicious time. It is cutting-edge technology in an underpenetrated market," added Shlomi Nachman, former Johnson & Johnson company group chairman of Cardiovascular & Specialty Solutions and Vision.
The ICE system, according to the company, addresses complex challenges related to peripheral thrombectomies. Eitan Konstantino, Ph.D., founder and CEO of Expanse Medical, said the ICE catheter system was engineered to harness the aspiration power found in a large bore catheter, within the slender profile of a much smaller device.
"It is clear this device was built with physicians in mind,” Dr. Michael Lichtenberg, chief medical officer of angiology at the Vascular Center Clinic in Arnsberg, Germany, told the press. “It aims to address some of the biggest issues we see regularly. The device was designed and built with a strong understanding of foundational physics that leaves me optimistic for its success.”
Expanse ICE is a company born from Expanse Medical, a medical device incubator founded by Dr. Konstantino. The team that built the Expanse ICE system also built AngioSculpt, Chocolate, and other neurovascular products. The company focuses on developing solutions for vascular diseases.
"I am looking forward to being among the first users of the Expanse ICE system. Its ability to offer powerful clot removal in a compact size is an intriguing proposition that could significantly enhance our ability to meet patient needs more effectively and improve treatment outcomes in peripheral vascular disease," said Dr. Aravinda Nanjundappa, director of peripheral vascular Interventions at the Cleveland Clinic.
"The thrombectomy market is witnessing robust growth and the approval of the ICE system comes at an auspicious time. It is cutting-edge technology in an underpenetrated market," added Shlomi Nachman, former Johnson & Johnson company group chairman of Cardiovascular & Specialty Solutions and Vision.