GenX Medical02.15.19
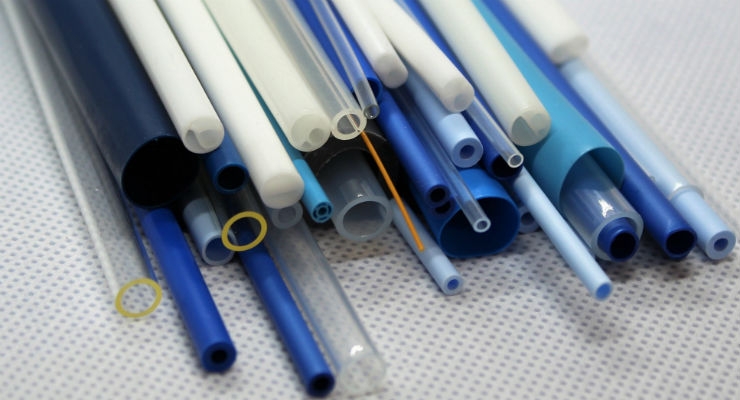
GenX Medical is a custom medical tubing supplier specializing in micro-bore tubing, bioresorbable extrusions, multi-lumen tubing, as well as other extrusions.
The company is pleased to announce they were recently awarded an ISO 13485:2016 Management System certificate for the manufacturer of medical grade extrusions intended for use in medical components by SRI Quality System Registrar. SRI is an internationally accredited registrar for management systems standards located in Pittsburgh, Pa.
According to Edward L. Maschmeier, associate vice president, certification, “We are pleased to acknowledge that GenX Medical LLC has demonstrated effective implementation of a management system. ISO 13485:2016 certification provides evidence to customers, suppliers, employees, and their community of their commitment to producing a quality product (service) and providing customer satisfaction.”
ISO 13485:2016 is process-based; certification recognizes organizations that can link business objectives with operating effectiveness. Companies that achieve Management System certification to ISO 13485:2016 have demonstrated effective implementation of documentation and records management, top management’s commitment to their customers, establishment of clear policy, good planning and implementation, good resource management, efficient process control, and measurement and analysis.
GenX Medical is known for its dedication to quick delivery of prototype quantities of custom medical extrusions. By securing many medical grade polymers, the company is able to ship custom tubing within only five to seven business days.
The company also designs and manufactures their own tooling dedicated to a client’s application; ensuring that the physical properties are customizable, repeatable and predictable between various lots.
“We truly believe that great business is done by building great relationships with our customers, our suppliers, and our employees, “ said Tyler Ware, president. “Those core values contribute to the significant growth we have seen over the last few years.”
In August of 2018, the company moved to a larger facility at in Chattanooga, Tenn. This facility is equipped with a controlled white room, and also boasts exceptional electrical utilities and additional space for future expansion.
The company is pleased to announce they were recently awarded an ISO 13485:2016 Management System certificate for the manufacturer of medical grade extrusions intended for use in medical components by SRI Quality System Registrar. SRI is an internationally accredited registrar for management systems standards located in Pittsburgh, Pa.
According to Edward L. Maschmeier, associate vice president, certification, “We are pleased to acknowledge that GenX Medical LLC has demonstrated effective implementation of a management system. ISO 13485:2016 certification provides evidence to customers, suppliers, employees, and their community of their commitment to producing a quality product (service) and providing customer satisfaction.”
ISO 13485:2016 is process-based; certification recognizes organizations that can link business objectives with operating effectiveness. Companies that achieve Management System certification to ISO 13485:2016 have demonstrated effective implementation of documentation and records management, top management’s commitment to their customers, establishment of clear policy, good planning and implementation, good resource management, efficient process control, and measurement and analysis.
GenX Medical is known for its dedication to quick delivery of prototype quantities of custom medical extrusions. By securing many medical grade polymers, the company is able to ship custom tubing within only five to seven business days.
The company also designs and manufactures their own tooling dedicated to a client’s application; ensuring that the physical properties are customizable, repeatable and predictable between various lots.
“We truly believe that great business is done by building great relationships with our customers, our suppliers, and our employees, “ said Tyler Ware, president. “Those core values contribute to the significant growth we have seen over the last few years.”
In August of 2018, the company moved to a larger facility at in Chattanooga, Tenn. This facility is equipped with a controlled white room, and also boasts exceptional electrical utilities and additional space for future expansion.