Niki Arrowsmith05.09.13
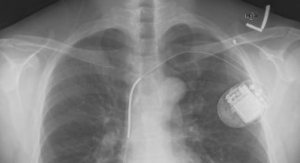
St. Paul, Minn.-based St. Jude Medical Inc. has received CE mark for its next next-generation Ellipse and SJM Assura portfolio of implantable cardioverter defibrillators (ICDs) and cardiac resynchronization therapy defibrillators (CRT-Ds). The new portfolio is designed to lower the risk for lead abrasion and to ensure high-voltage therapy delivery.
“The goal for this innovative portfolio of devices is to bring implantable defibrillation reliability and patient safety to the next level,” said Eric S. Fain, M.D., president of the St. Jude’s Implantable Electronic Systems division. “The new Ellipse ICD and Assura family of devices demonstrates St. Jude Medical’s commitment to developing technologies that provide physicians with advanced patient management tools to mitigate the most common ICD lead complications, especially those that can lead to ineffective high voltage therapy delivery.”
The Ellipse and SJM Assura family of devices feature the Dynamictxm over-current detection algorithm, which works by automatically adjusting shocking configurations within the device to ensure the delivery of high-voltage therapy if an electrical short in one portion of the system were to occur. The portfolio of implantable defibrillators features low friction coating on the device can, which has been demonstrated in testing to reduce the friction between the device and leads. As such, the low-friction coating is designed to reduce the risk for lead-to-can abrasion, the most common type of lead insulation failure in the industry.
St. Jude has faced controversy over failed defibrillator leads in the past. Last year, the company’s Riata leads were accused of causing 22 patient deaths, due to frayed leads caused by excessive friction.
The reduced friction technologies are hoped to provide preventative and adaptive capabilities to address potential failures that can result in the inability to deliver high voltage therapy when needed, especially in systems using silicone-only insulated defibrillation leads, which are, according to St. Jude, known to be at higher risk of abrasion. It is estimated that over 400,000 silicone-only insulated defibrillation leads from all manufacturers remain active worldwide.
“The annual rate of lead defects increases over time regardless of manufacturer and insulation failure accounts for 70 percent of lead failures in older leads,” said Johan Vijgen, M.D., director of electrophysiology at Jessa Hospitals in Hasselt, Belgium. “With important safety features designed to ensure successful delivery of high-voltage therapy, these new devices offer my patients a higher standard of care.”
The Ellipse and SJM Assura family of devices also feature expanded protection against inappropriate and unnecessary shocks with Securesense RV lead noise discrimination, an algorithm that expands St. Jude Medical Shockguard Technology. Securesense RV lead noise discrimination is designed to differentiate lead noise (over-sensing of electrical signals) from true ventricular tachycardia or ventricular fibrillation episodes requiring therapy.
The St. Jude Medical high-voltage portfolio is augmented by the Durata defibrillation lead with Optim lead insulation, onto which the company shifted more focus after the Riata lead debacle.
The Ellipse ICD delivers 36 joules in delivered energy, providing a downsized alternative to the Fortify Assura ICD, purportedly without compromising on energy, longevity, safety or reliability.
The Assura family of devices allows St. Jude Medical to continue to offer 40 joules in delivered energy and includes the Quadra Assura CRT-D, the Unify Assura CRT-D and the Fortify Assura ICD.
St. Jude Medical is provides a broad range of medical devices and technology in the cardiac, neurological and chronic pain spaces.
“The goal for this innovative portfolio of devices is to bring implantable defibrillation reliability and patient safety to the next level,” said Eric S. Fain, M.D., president of the St. Jude’s Implantable Electronic Systems division. “The new Ellipse ICD and Assura family of devices demonstrates St. Jude Medical’s commitment to developing technologies that provide physicians with advanced patient management tools to mitigate the most common ICD lead complications, especially those that can lead to ineffective high voltage therapy delivery.”
The Ellipse and SJM Assura family of devices feature the Dynamictxm over-current detection algorithm, which works by automatically adjusting shocking configurations within the device to ensure the delivery of high-voltage therapy if an electrical short in one portion of the system were to occur. The portfolio of implantable defibrillators features low friction coating on the device can, which has been demonstrated in testing to reduce the friction between the device and leads. As such, the low-friction coating is designed to reduce the risk for lead-to-can abrasion, the most common type of lead insulation failure in the industry.
St. Jude has faced controversy over failed defibrillator leads in the past. Last year, the company’s Riata leads were accused of causing 22 patient deaths, due to frayed leads caused by excessive friction.
The reduced friction technologies are hoped to provide preventative and adaptive capabilities to address potential failures that can result in the inability to deliver high voltage therapy when needed, especially in systems using silicone-only insulated defibrillation leads, which are, according to St. Jude, known to be at higher risk of abrasion. It is estimated that over 400,000 silicone-only insulated defibrillation leads from all manufacturers remain active worldwide.
“The annual rate of lead defects increases over time regardless of manufacturer and insulation failure accounts for 70 percent of lead failures in older leads,” said Johan Vijgen, M.D., director of electrophysiology at Jessa Hospitals in Hasselt, Belgium. “With important safety features designed to ensure successful delivery of high-voltage therapy, these new devices offer my patients a higher standard of care.”
The Ellipse and SJM Assura family of devices also feature expanded protection against inappropriate and unnecessary shocks with Securesense RV lead noise discrimination, an algorithm that expands St. Jude Medical Shockguard Technology. Securesense RV lead noise discrimination is designed to differentiate lead noise (over-sensing of electrical signals) from true ventricular tachycardia or ventricular fibrillation episodes requiring therapy.
The St. Jude Medical high-voltage portfolio is augmented by the Durata defibrillation lead with Optim lead insulation, onto which the company shifted more focus after the Riata lead debacle.
The Ellipse ICD delivers 36 joules in delivered energy, providing a downsized alternative to the Fortify Assura ICD, purportedly without compromising on energy, longevity, safety or reliability.
The Assura family of devices allows St. Jude Medical to continue to offer 40 joules in delivered energy and includes the Quadra Assura CRT-D, the Unify Assura CRT-D and the Fortify Assura ICD.
St. Jude Medical is provides a broad range of medical devices and technology in the cardiac, neurological and chronic pain spaces.