In the rapidly evolving global landscape of healthcare industries, the quest for innovative materials has never been more critical. Among the myriad of materials available, synthetic polyisoprene and silicone elastomers stand out as two remarkable contenders, offering a plethora of unique attributes that hold immense promise for advancing medical and surgical device applications, especially where safety, reliability, and patient comfort are paramount.
Indeed, the success or failure of a new medical device can depend on material selection. This is especially true for elastomers, which are far more complex and variable than most other medical materials. In fact, improper material selection is the leading cause of elastomeric product failure.

Brian Highley, CEO, Cirtec Medical
This Q&A with Dr. Amalendu Sarkar, VP of materials technology at Cirtec Medical, aims to explore and elucidate some of those distinctive features that make synthetic polyisoprene and silicone elastomers exceptional choices in healthcare contexts, and it may help design engineers choose their path for selection of the right elastomeric materials for their next generation, innovative medical and surgical devices.
Sean Fenske: What makes synthetic polyisoprene and silicone elastomers unique as compared to other elastomer materials.
Dr. Amalendu Sarkar: Synthetic polyisoprene Elastomer (IR) stands out from other elastomer materials due to its distinct combination of properties and characteristics. These unique qualities contribute to its widespread use in various industries—from medical to automotive—where performance, reliability, and versatility are essential. Synthetic polyisoprene replicates the molecular structure of natural rubber (NR) without the presence of NR’s organic proteins and impurities from non-rubber constituents. This gives IR the desirable physical and mechanical properties of NR while eliminating its allergens.
- Elastic recovery: Like NR, synthetic IR with higher Cis-1,4-content (which is also NR’s foundational structure) demonstrates strain induced crystallization (SIC)—the ability to strengthen when stretched. This unique characteristic empowers synthetic Cis-1,4 polyisoprene with enhanced tensile and tear strength properties, making it an excellent choice for applications demanding exceptional durability.
- Barrier properties: In the medical industry, synthetic polyisoprene garners notable recognition for its outstanding resealability. This characteristic enables synthetic polyisoprene to maintain the critical barrier properties essential in septum, stopper, or closure applications, even after repeated needle punctures. Furthermore, a correctly formulated polyisoprene septum or stopper will remain free from coring (the shedding of material fragments) after repeated puncture. These attributes make synthetic polyisoprene an unmatched and preferred choice for various fluid management applications in the medical field.
On the other hand, silicone is inextricably linked with numerous medical devices. The strong, highly stable silicone-oxygen bonds in its main chain backbone set it apart from polyisoprene and other organic elastomers that rely on carbon-carbon bonds. This molecular architecture bestows silicone with exceptional stability, flexibility, and biological inertness. These attributes are of paramount importance in healthcare applications, offering unrivaled biocompatibility, an extensive operational temperature range, compatibility with sterilization processes, remarkable flexibility, softness, transparency, clarity, durability, and extended shelf-life. These unique qualities make silicone a material of choice for a wide array of medical applications.
Fenske: For what types of medical applications are these materials typically used?
Dr. Sarkar: Both silicone and synthetic polyisoprene elastomer products find extensive use in various medical applications due to their unique combination of properties that make them well-suited to ensure patient safety, comfort, and effectiveness.
Common products produced with synthetic polyisoprene elastomer include:
- Seals
- Slitted Valves
- Bellows
- Duckbills
- O-Rings
- Rubber Stoppers
- Septum for IV Systems
- Valve Components
- Diaphragms
- Plunger Tips
- Needle Shields
- Resealable Components in Surgical Settings
- Drug Delivery Products
- Fluids Management Components
Cirtec Medical offers drug master filed (DMF) polyisoprene materials, catering to diverse healthcare needs.
On the other hand, silicone products have gained prominence in the modern healthcare field due to their exceptional combination of properties that make them uniquely suitable for a variety of medical applications, including:
-
Medical Implants: Silicone is used to create a wide range of medical implants, including breast implants, cochlear implants, intraocular lenses, and integral components of implantable pulse generators (IPGs). I would like to add that Cirtec Medical has a strong presence in the specialty IPG product market, which includes devices for neuromodulation and active implants.
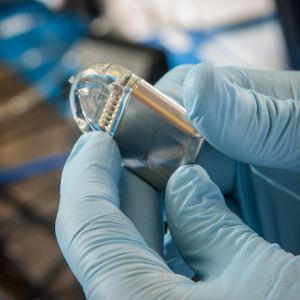
Silicone is frequently used to create integral components for medical implants such as IPGs. - Medical Tubing and Catheters: Silicone is an ideal material for medical tubing and catheters due to its flexibility, biocompatibility, low extractables and leachables, and reduced interactivity with bodily fluids. It is used in applications such as intravenous (IV) lines, urinary catheters, breast pumps, enteral tubes, peristaltic pumps, and drainage tubes.
- Surgical Instruments and Tools: Silicone is employed to create ergonomic grips and handles for surgical instruments. Its non-slip properties enhance the precision and comfort of surgical procedures.
- Medical Seals and Gaskets: Silicone is utilized to manufacture seals and gaskets for medical devices and equipment, ensuring proper function and sterility. These components are commonly used in pumps, valves, and respiratory devices.
- Wound Dressings and Adhesives: Silicone is used to create wound dressings and adhesive products that are gentle on the skin and provide a desired protective barrier for wounds. Its non-stick, non-allergic properties help prevent adherence to wounds during the healing process.
- Respiratory Equipment: Silicone is employed in the creation of components for respiratory equipment such as masks, tubing, and valves. Its biocompatibility and resistance to sterilization processes are critical for maintaining patient safety.
- Dental Applications: In dentistry, silicone is utilized to create dental impression materials, bite guards, dental molds, and orthodontic devices. Its accuracy and stability during dental procedures make it a preferred material of choice.
- Medical Balloons: Silicone is utilized in the production of medical balloons used in products like angioplasty, catheterization, and dilation. Its ability to expand while maintaining surgical integrity is crucial for these applications.
- Prosthetics and Orthotics: The flexibility, durability, and softness of silicone makes it extremely suitable for prosthetics, orthotics, and custom-made medical devices. It helps create comfortable and functional solutions for patients with mobility challenges.
- Dialysis Equipment: Silicone is used in dialysis equipment components due to its compatibility with blood-contact applications and resistance to chemicals used in dialysis procedures.
Both synthetic polyisoprene and silicone elastomers play a crucial role in enhancing patient care, improving medical device performance, and contributing to innovative healthcare solutions.
Fenske: What benefits do they offer to medical device manufacturers such that they are a preferred material?
Dr. Sarkar: Both synthetic polyisoprene and silicone elastomers offer a wide range of benefits to medical device manufacturers, making them a preferred material for various applications within the medical field. These advantages contribute to the materials’ properties and its widespread use in medical device manufacturing.
The major benefit of synthetic IR is its high green strength, good hysteresis and elastic recovery properties, extremely good flex, and dynamic properties. There is a unique phenomenon in synthetic IR material called strain induced crystallization (SIC) that helps to resist the crack initiation and propagation. It has extremely good tensile and tear strength properties. Properly formulated polyisoprene material demonstrates high resealability characteristics with no coring phenomenon. Synthetic polyisoprene is also relatively less permeable to various gases.
Well-crafted silicone is highly biocompatible, which means it is well-tolerated by the human body and doesn’t trigger any adverse reactions. This unique property alone is essential for some medical and healthcare products that come into direct and prolonged contact with bodily tissues and fluids, ensuring patient safety and minimizing the risk of complications. Being a well-documented and well-established biocompatible and bio-durable material, silicone is considered as one of the most widely used polymeric materials in healthcare markets. It is also extremely stable in the presence of UV-light material. It is easily sterilizable by various techniques such as EtO, gamma, e-beam radiation, steam, ozone, etc.
Fenske: What challenges do these materials present that can make them difficult to use within a medical device? How do you overcome these?
Dr. Sarkar: While synthetic polyisoprene materials offer numerous advantages for medical device applications, they can also pose certain challenges that need to be addressed to ensure their effectiveness and safe use. Following are some key challenges associated with synthetic polyisoprene materials in medical devices.
- Additives in Compounding: In general, gum synthetic polyisoprene elastomer cannot be used as such for any practical purpose. Different additives such as antioxidants and curatives are indispensable parts of rubber compounding. Although they are added in smaller amounts, selection and optimization of those ingredients are highly critical. Ideally, it is preferable that the material should be free from polynuclear aromatics (PNAs) and nitrosamine.
- Silicone Elastomer Challenges: Silicone, while versatile, can be relatively more expensive and in some special applications, it is reported to absorb proteins and antioxidants, which reduces the efficacy of the drug solutions.
To address these challenges and harness the advantages of synthetic polyisoprene and silicone elastomers for the development of safe, reliable, and effective medical devices, rigorous testing and collaboration with experts experienced in using these medical materials and adhering to regulatory guidelines are strongly recommended for medical device manufacturers. By navigating these challenges effectively, manufacturers can unlock the full potential of these materials while ensuring the quality and safety of their products.
Fenske: Where are the growth areas for these materials? What use cases are being explored that are outside the "norm" for these materials within medical device manufacturing?
Dr. Sarkar: Both synthetic polyisoprene and silicone materials are finding significant growth opportunities and are being explored for innovative use cases across various domains of medical device manufacturing, extending beyond conventional applications.
As mentioned previously, synthetic polyisoprene materials are growing in the medical application segment such as Septa, slitted and un-slitted valves, seals, thin wall bellows, sleeve stoppers, O-rings, large and small volume parenteral stoppers, diaphragms, IV components, needle shield, resealable insulin plugs, among others.
On the other hand, silicone products are witnessing growth across a wide spectrum of medical segments. These encompass general surgery, cardiology, oncology, ophthalmology, orthopedics, over-molded surgical products, slitted valves, catheter tubes, peristaltic pumps, medical cables, dental impression segments, respiratory device industries, and implantable products, to name a few.
The continuous expansion of these materials into diverse medical applications underscores their versatility and adaptability, as well as their ability to meet evolving healthcare needs and drive innovation within the medical device manufacturing sector.
Fenske: What are misconceptions associated with synthetic polyisoprene and silicone elastomers? What do you say to clear these up?
Dr. Sarkar: Misconceptions about synthetic polyisoprene and silicone elastomers can arise due to misunderstanding or incomplete information. Following are some common misconceptions that need clarification:
- Synthetic polyisoprene is the same as natural rubber latex (NRL): Synthetic polyisoprene is a distinct material synthesized from petroleum sources to mimic the properties of natural rubber while reducing the risk of allergenic reactions and improve consistency. Synthetic polyisoprene offers enhanced biocompatibility and no allergenic potential like NRL. It is worth mentioning Cirtec Medical offers quite a few cost-effective polyisoprene materials that have a long history of use in various surgical devices, drug delivery applications, and healthcare applications.
- Silicone is toxic and harmful to the body: High quality medical grade silicone rubber is biocompatible and extensively tested for safety. It meets stringent regulatory standards for medical device applications. Medical-grade silicone is inert, non-reactive, and hypoallergenic. Some silicone materials have a long history of safe use in medical devices and have been proven suitable for both short- and long-term implantable products.
- Silicone elastomer materials are expensive: While high-quality materials may come at a cost, the overall value they provide in terms of biocompatibility, bio-durability, and overall performance, in many cases, outweighs the initial cost. We definitely need to have a look at the cost of material, but we also need to have a look at the cost of failure depending on the criticality of the end application.
Fenske: Do you have any additional comments you'd like to share based on any topics we discussed or something you'd like to share based on any of the topics we discussed or something you'd like to tell mechanical device manufacturers?
Dr. Sarkar: Certainly! We would like to offer a few additional insights and advice for medical device manufacturers based on all the topics discussed in response to other questions in this interview.
The choice of elastomeric materials is critical in medical device manufacturing. When selecting synthetic polyisoprene or silicone elastomers, consider the specific requirements of your device, including biocompatibility, mechanical properties, resealability characteristics, sterilization compatibility, and duration of performance in actual application. Collaborate with material experts for innovative solutions, interdisciplinary teams, and design engineers to ensure you’re using the most suitable material for your application. Keeping the patient at the center of your device development efforts is key to success. As the industry evolves, consider the environmental impact of your devices and materials as well. Explore ways to minimize waste, reduce energy consumption, and choose materials that align with sustainable practices.
In a rapidly advancing field like medical device manufacturing, staying informed, collaborative, and adaptable is crucial. By harnessing the unique properties of synthetic polyisoprene and silicone elastomers, and by fostering innovation and patient-centricity, manufacturers can contribute to transformative advancements in healthcare technology and, ultimately, improve patient lives.
As a vertically integrated, full-service outsource partner, Cirtec Medical has decades of expertise to excel in every phase of the product development lifecycle for complex medical devices. Our core capabilities encompass advanced material formulations, design, development, and manufacturing, allowing us to deliver comprehensive solutions. At Cirtec Medical, our experienced team specializes in tackling even the most formidable manufacturing challenges.
If you find yourself in need of a solution or facing difficulties in creating a device or component, we encourage you to leverage the expertise of our team. Collaborating with your team, we aim to provide the best possible solutions tailored to your specific application.
Click here to find out more about Cirtec Medical >>>>>













