Russell M. Singleton, Ph.D., President, Russ Singleton Consulting LLC; Aaron Joseph, Principal Consultant, Sunstone Pilot Inc.04.22.24
This marks the start of a series of columns in which we will describe a set of concepts and methods for managing the development of complex medical devices. We start by explaining the particular challenges of developing complex medical devices and why they need to be managed differently than other medical devices.
The (fictional) Eagle Project, aiming to develop a complex surgical robotic system, faces a critical delay issue. The CEO is frustrated by the repeated setbacks undermining the planned product launch. The company has successfully developed simpler medical devices already. Why is this project different?
This new surgical robotic system was the first time the company had developed a truly complex product. They knew it would cost more to develop and had budgeted accordingly. However, they didn’t anticipate the particular challenges of developing a complex system and didn’t adjust their product development approach. Their approach, which was successful in developing single-use medical devices, was overmatched by the challenges of developing a significantly more complex medical device.
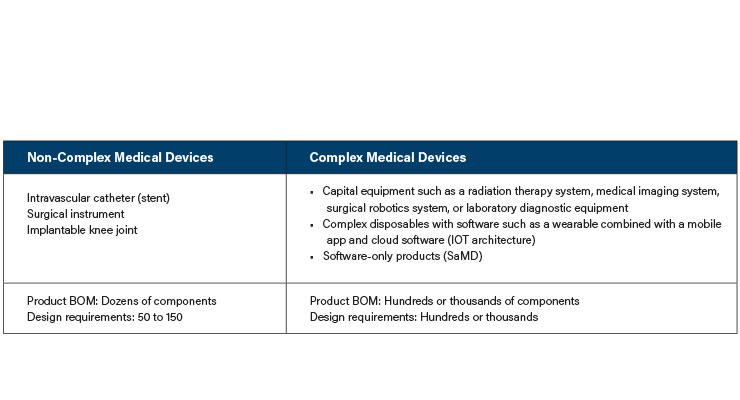
In this series of columns, we will examine what is different about the development of complex medical devices, how project risks are multiplied, and why different methods are required to maximize the chance of successful development. This discussion focuses on new product development (NPD) of complex medical devices and especially how to avoid traps that teams may fall into. By “new,” we are distinctly separating the kind of development that is incremental or “me too” in nature and focusing on products that involve new applications, technology, or science. By “complex,” we generally mean development that includes hardware, software, and perhaps other modalities such as optics (Table 1). Further, these development projects require a large team of individuals from diverse specialties working together to achieve an outcome. The issues we discuss may crop up with simpler medical devices or non-medical products, but they are particularly challenging for complex medical devices.
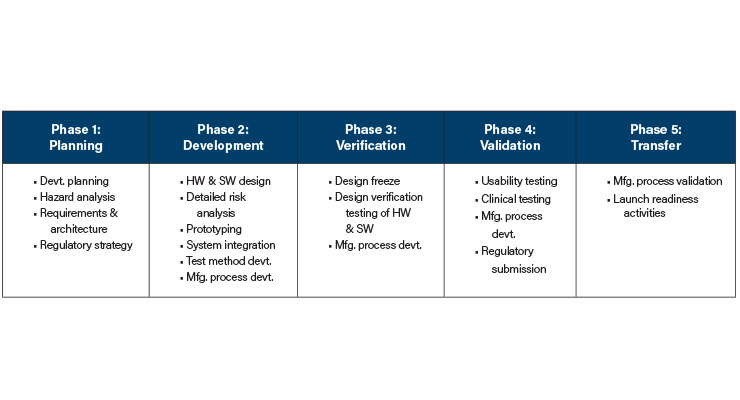
The product team followed the company’s well-established product development process consisting of five phases (Table 2). With this process, the team conducts a design review at the end of each phase using a detailed checklist defining exactly the required documents to be completed for the phase. This detailed framework is intended to ensure every project completes all necessary deliverables, complies with all applicable regulations and standards, and stays on schedule. The Eagle product team completed Phase 1: Planning successfully on time.
However, the project encountered significant setbacks after Phase 1. The anticipated two-week system integration phase stretched into a 10-month ordeal due to persistent issues integrating hardware and software components. After finally achieving a working prototype, usability testing revealed serious shortcomings—ergonomic flaws and the lack of some important features that surgeons expected. This discovery prompted contentious redesign meetings, costing the company additional time and money. Design engineers, who had moved on to other projects, were brought back to address the issues, exacerbating delays. Adding to the frustration and delays was a mountain of documentation that had to be painstakingly updated and re-released to capture all the changes.
As these challenges mounted, the CEO grew concerned that the Eagle Project's protracted issues could jeopardize the company's broader product launch roadmap with a cascade of delays. The project, initially a beacon of hope, had turned into a costly and time-consuming ordeal.
The problems of the Eagle Project are not unique. We have seen many of the same problems across dozens of NPD projects:
Russ Singleton, Ph.D., president of Russ Singleton Consulting LLC, is a consultant based in California. He has extensive experience in VP R&D, general management, and C-suite roles in the semiconductor equipment and medtech sectors. He has successfully transform-ed development teams in inspection systems, DNA sequencing, and various medical imaging and surgical robotics systems. Singleton has a Ph.D. and M.S. in electrical engineering from the University of Illinois and a bachelor of engineering from the Pratt Institute.
Aaron Joseph, principal consultant with Sunstone Pilot Inc., is a biomedical engineer based in Waltham, Mass. He has over 20 years of experience in medical device development across a broad range of products: surgical robotics systems, laser eye surgery equipment, wearables, X-ray imaging systems, drug inhaler devices, catheters, and multiple IOT and SaMD products. He helps clients with risk management and design controls, software validation, training, and implementation of software tools for documentation automation.
The (fictional) Eagle Project, aiming to develop a complex surgical robotic system, faces a critical delay issue. The CEO is frustrated by the repeated setbacks undermining the planned product launch. The company has successfully developed simpler medical devices already. Why is this project different?
A Project in Crisis
There was a deafening silence in the conference room. The project manager had just announced to the executive team that the Eagle Project—to develop a new surgical robotic system—would be delayed by another six months. The silence was finally broken by the CEO shouting, “This is unacceptable! I promised the board that we would ship the product by Q2 of next year. We cannot delay the Eagle Project again!” The CEO didn’t know the product team had spent the previous week desperately trying to figure out a way to minimize the delay. Unfortunately, the roots of this problem started three years earlier when the project began. The hapless project manager could only pursue a costly fix to the new robotic system. What caused this latest project delay and the previous delays? The company had already successfully developed and commercialized previous medical devices. Why was this project such a disaster?This new surgical robotic system was the first time the company had developed a truly complex product. They knew it would cost more to develop and had budgeted accordingly. However, they didn’t anticipate the particular challenges of developing a complex system and didn’t adjust their product development approach. Their approach, which was successful in developing single-use medical devices, was overmatched by the challenges of developing a significantly more complex medical device.
In this series of columns, we will examine what is different about the development of complex medical devices, how project risks are multiplied, and why different methods are required to maximize the chance of successful development. This discussion focuses on new product development (NPD) of complex medical devices and especially how to avoid traps that teams may fall into. By “new,” we are distinctly separating the kind of development that is incremental or “me too” in nature and focusing on products that involve new applications, technology, or science. By “complex,” we generally mean development that includes hardware, software, and perhaps other modalities such as optics (Table 1). Further, these development projects require a large team of individuals from diverse specialties working together to achieve an outcome. The issues we discuss may crop up with simpler medical devices or non-medical products, but they are particularly challenging for complex medical devices.
Common Problems and Causes
The Eagle Project initially showed promise as a team of nearly 40 people, led by an experienced project leader and guided by a retired surgeon, embarked on developing a new surgical robotic system. They efficiently defined product requirements and organized the work into electrical, mechanical, software, and manufacturing streams. A detailed project schedule was established, aiming to culminate in a system integration milestone followed by verification and validation testing and transfer to manufacturing.The product team followed the company’s well-established product development process consisting of five phases (Table 2). With this process, the team conducts a design review at the end of each phase using a detailed checklist defining exactly the required documents to be completed for the phase. This detailed framework is intended to ensure every project completes all necessary deliverables, complies with all applicable regulations and standards, and stays on schedule. The Eagle product team completed Phase 1: Planning successfully on time.
However, the project encountered significant setbacks after Phase 1. The anticipated two-week system integration phase stretched into a 10-month ordeal due to persistent issues integrating hardware and software components. After finally achieving a working prototype, usability testing revealed serious shortcomings—ergonomic flaws and the lack of some important features that surgeons expected. This discovery prompted contentious redesign meetings, costing the company additional time and money. Design engineers, who had moved on to other projects, were brought back to address the issues, exacerbating delays. Adding to the frustration and delays was a mountain of documentation that had to be painstakingly updated and re-released to capture all the changes.
As these challenges mounted, the CEO grew concerned that the Eagle Project's protracted issues could jeopardize the company's broader product launch roadmap with a cascade of delays. The project, initially a beacon of hope, had turned into a costly and time-consuming ordeal.
The problems of the Eagle Project are not unique. We have seen many of the same problems across dozens of NPD projects:
- Launching directly into development without much or any customer insight
- Waiting to truly understand the customer’s needs until a working product is ready for evaluation
- Discovering design flaws late in development, leading to expensive delays
- Schedule overruns with software development
- Underestimating the development time from a working breadboard to a commercial product (just a matter of implementation)
- Selecting a project lead who has product development experience with non-complex medical devices but has no systems experience or insight
- Planning only a single major integration milestone to combine subsystems developed in silos
- Lackluster sales of a new product after launch without a good understanding of why
- Compliance problems with meeting design controls (one of the most common FDA audit findings)
Russ Singleton, Ph.D., president of Russ Singleton Consulting LLC, is a consultant based in California. He has extensive experience in VP R&D, general management, and C-suite roles in the semiconductor equipment and medtech sectors. He has successfully transform-ed development teams in inspection systems, DNA sequencing, and various medical imaging and surgical robotics systems. Singleton has a Ph.D. and M.S. in electrical engineering from the University of Illinois and a bachelor of engineering from the Pratt Institute.
Aaron Joseph, principal consultant with Sunstone Pilot Inc., is a biomedical engineer based in Waltham, Mass. He has over 20 years of experience in medical device development across a broad range of products: surgical robotics systems, laser eye surgery equipment, wearables, X-ray imaging systems, drug inhaler devices, catheters, and multiple IOT and SaMD products. He helps clients with risk management and design controls, software validation, training, and implementation of software tools for documentation automation.