Sam Brusco, Associate Editor10.05.17
Star Trek’s replicator isn’t looking so far-fetched these days. For those unfamiliar, the classic sci-fi odyssey’s transporter technology materialized food, spare parts, and clothing for the Enterprise crew at a moment’s notice. Starry-eyed readers may notice the similarity between that device and a manufacturing method that has peppered popular culture with headlines in reality—the 3D printer. The machines even resemble replicators, and have printed both food and spare jet parts for GE Aviation projects.
The next item on the printer’s queue? Biomaterial products that can function alongside living cells, or “bio-inks.” Bio-inks are typically composed of laboratory-grown cells immersed in a hydrogel scaffold, which is a water-swollen polymeric material that maintains a distinct three-dimensional structure. These serve as the building blocks for bioprinting, or assembling the bio-ink layer-by-layer into specific organ or tissue shapes. The approach could revolutionize regenerative medicine by offering a reliable way to manufacture complex tissues for support, repair, or augmentation of diseased and damaged areas of the body.
Producing tissues this way still faces some issues, however. It’s difficult to control the position of cells in 3D because they often move within printed constructs and the hydrogel scaffold supporting the cells can collapse in on itself. Conventional hydrogels also fall prey to thermal instability—even minute temperature changes can drastically alter the construct’s viscosity.
“This makes it problematic for many room temperature biofabrication systems,” said Keekyoung Kim, an assistant professor at UBC Okanagan’s School of Engineering, whose team developed a new bio-ink in mid-September. “These are compatible with only a narrow range of hydrogel viscosities and must generate products that are as uniform as possible if they are to function properly.”
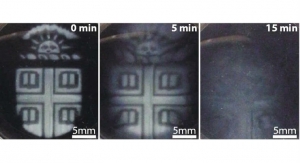
To tackle the instability problem, Kim’s team first analyzed the physical and biological properties of three different gelatin methacrylate (GelMA) hydrogels: porcine skin, cold-water fish skin, and cold-soluble gelatin, which is primarily used in culinary projects. It was found hydrogel made from cold-soluble gelatin performed best and offered a strong candidacy for future 3D organ printing. Cold-soluble gelatin, very fortunately, dissolves without heat and is three times cheaper than porcine skin. The cold-soluble GelMA was shown to form healthy tissue scaffolds, allowed cells to grow and adhere to it, and was thermally stable and formed consistently uniform droplets at room temperature.
“We hope this new bio-ink will help researchers create improved artificial organs and lead to the development of better drugs, tissue engineering, and regenerative therapies,” Kim said. “The next step is to investigate whether or not cold-soluble GelMA-based tissue scaffolds can be used long-term both in the laboratory and in real-world transplants.”
Researchers abroad are also developing new methods to print laboratory grown cells into complex tissues by making the scaffolding more stable. A team comprised of University of Oxford’s Departments of Chemistry and Physiology, Anatomy, and Genetics as well as scientists from the Centre for Molecular Medicine at Bristol devised a way to make tissues in self-contained cells that retain their shape.
The team immersed lab-grown cells in protective nanoliter droplets, which were then wrapped in a lipid coating. The resulting bio-ink was assembled, layer-by-layer, into a living structure. The researchers found the cells’ survival rate was improved, allowing the team to build each tissue one drop at a time to the desired resolution. This method made fabrication of patterned cell constructs possible. Once fully grown, these resemble or potentially augment natural tissues—a necessary component for their utility in tissue engineering.
“We were aiming to fabricate three-dimensional living tissues that could display the basic behaviors and physiology found in natural organisms,” explained Dr. Alexander Graham, lead author of the study and 3D Bioprinting Scientist at OxSyBio (Oxford Synthetic Biology). “To date, there are limited examples of printed tissues, which have the complex cellular architecture of native tissues. Hence, we focused on designing a high-resolution cell printing platform, from relatively inexpensive components, that could be used to reproducibly produce artificial tissues with appropriate complexity from a range of cells including stem cells.”
The team finished its research in 2016, and since then OxSyBio has been spun-out from the University of Oxford laboratory. The company seeks to commercialize its technique for both industrial and biomedical purposes. In the coming months, the firm will develop new complementary printing techniques allowing a wider range of living and hybrid materials in order to make tissues at an industrial scale.
“There are many potential applications for bioprinting, and we believe it will be possible to create personalized treatments by using cells sourced from patients to mimic or enhance natural tissue function,” commented Dr. Sam Olof, chief technology officer at OxSyBio. “In the future, 3D bioprinted tissues maybe also be used for diagnostic applications—for example, for drug or toxin screening.”
“The bioprinting approach developed with Oxford University is very exciting, as the cellular constructs can be printed efficiently at extremely high resolution with very little waste,” said Dr. Adam Perriman, a researcher at the University of Bristol’s School of Cellular and Molecular Medicine. “The ability to 3D print with adult stem cells and still have them differentiate was remarkable, and really shows the potential of this new methodology to impact regenerative medicine globally.”
The next item on the printer’s queue? Biomaterial products that can function alongside living cells, or “bio-inks.” Bio-inks are typically composed of laboratory-grown cells immersed in a hydrogel scaffold, which is a water-swollen polymeric material that maintains a distinct three-dimensional structure. These serve as the building blocks for bioprinting, or assembling the bio-ink layer-by-layer into specific organ or tissue shapes. The approach could revolutionize regenerative medicine by offering a reliable way to manufacture complex tissues for support, repair, or augmentation of diseased and damaged areas of the body.
Producing tissues this way still faces some issues, however. It’s difficult to control the position of cells in 3D because they often move within printed constructs and the hydrogel scaffold supporting the cells can collapse in on itself. Conventional hydrogels also fall prey to thermal instability—even minute temperature changes can drastically alter the construct’s viscosity.
“This makes it problematic for many room temperature biofabrication systems,” said Keekyoung Kim, an assistant professor at UBC Okanagan’s School of Engineering, whose team developed a new bio-ink in mid-September. “These are compatible with only a narrow range of hydrogel viscosities and must generate products that are as uniform as possible if they are to function properly.”
To tackle the instability problem, Kim’s team first analyzed the physical and biological properties of three different gelatin methacrylate (GelMA) hydrogels: porcine skin, cold-water fish skin, and cold-soluble gelatin, which is primarily used in culinary projects. It was found hydrogel made from cold-soluble gelatin performed best and offered a strong candidacy for future 3D organ printing. Cold-soluble gelatin, very fortunately, dissolves without heat and is three times cheaper than porcine skin. The cold-soluble GelMA was shown to form healthy tissue scaffolds, allowed cells to grow and adhere to it, and was thermally stable and formed consistently uniform droplets at room temperature.
“We hope this new bio-ink will help researchers create improved artificial organs and lead to the development of better drugs, tissue engineering, and regenerative therapies,” Kim said. “The next step is to investigate whether or not cold-soluble GelMA-based tissue scaffolds can be used long-term both in the laboratory and in real-world transplants.”
Researchers abroad are also developing new methods to print laboratory grown cells into complex tissues by making the scaffolding more stable. A team comprised of University of Oxford’s Departments of Chemistry and Physiology, Anatomy, and Genetics as well as scientists from the Centre for Molecular Medicine at Bristol devised a way to make tissues in self-contained cells that retain their shape.
The team immersed lab-grown cells in protective nanoliter droplets, which were then wrapped in a lipid coating. The resulting bio-ink was assembled, layer-by-layer, into a living structure. The researchers found the cells’ survival rate was improved, allowing the team to build each tissue one drop at a time to the desired resolution. This method made fabrication of patterned cell constructs possible. Once fully grown, these resemble or potentially augment natural tissues—a necessary component for their utility in tissue engineering.
“We were aiming to fabricate three-dimensional living tissues that could display the basic behaviors and physiology found in natural organisms,” explained Dr. Alexander Graham, lead author of the study and 3D Bioprinting Scientist at OxSyBio (Oxford Synthetic Biology). “To date, there are limited examples of printed tissues, which have the complex cellular architecture of native tissues. Hence, we focused on designing a high-resolution cell printing platform, from relatively inexpensive components, that could be used to reproducibly produce artificial tissues with appropriate complexity from a range of cells including stem cells.”
The team finished its research in 2016, and since then OxSyBio has been spun-out from the University of Oxford laboratory. The company seeks to commercialize its technique for both industrial and biomedical purposes. In the coming months, the firm will develop new complementary printing techniques allowing a wider range of living and hybrid materials in order to make tissues at an industrial scale.
“There are many potential applications for bioprinting, and we believe it will be possible to create personalized treatments by using cells sourced from patients to mimic or enhance natural tissue function,” commented Dr. Sam Olof, chief technology officer at OxSyBio. “In the future, 3D bioprinted tissues maybe also be used for diagnostic applications—for example, for drug or toxin screening.”
“The bioprinting approach developed with Oxford University is very exciting, as the cellular constructs can be printed efficiently at extremely high resolution with very little waste,” said Dr. Adam Perriman, a researcher at the University of Bristol’s School of Cellular and Molecular Medicine. “The ability to 3D print with adult stem cells and still have them differentiate was remarkable, and really shows the potential of this new methodology to impact regenerative medicine globally.”