Sam Brusco, Associate Editor05.01.17
Scientists are beginning to run out of superlatives for graphene, the two-dimensional wonder material. It’s been hailed as the lightest, strongest, thinnest, and best heat- and electricity-conducting material ever discovered. Similar to the plastics revolution of the 20th century, the carbon allotrope seems eagerly poised to usher in “the age of graphene” in the 21st century. (For more information on graphene and other medical device material trends, check out my feature articles in the March edition of MPO and the March/April edition of MPO’s sister publication Orthopedic Design & Technology, entitled “Go Out There and Show Them What You’re Made Of” and “Made of Sterner Stuff,” respectively.)
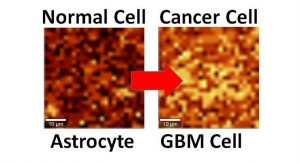
For a few years now, graphene has been tested for an impressive arsenal of futuristic medical technologies. It’s been researched as a platform to differentiate normal from cancerous cells, a replacement for titanium as a bone-filler, a nanoribbon to reconnect damaged spinal cords, and a strain sensor after insertion into silly putty.
Graphene-based electronics have the most promise in vaulting the material into the mainstream due to the its astounding electrical conductivity. Graphene assumes a flat, hexagonal lattice structure, which allows relatively low resistance to the flow of electrons. It fares better as a conductor than copper, reaching almost superconductor levels of power. Specifically, in its lattice form, graphene is known to be a “zero-gap semiconductor,” meaning its conduction and valence bonds meet at the Dirac points (six locations in momentum space on the edge of the Brillouin zone, divided into two non-equivalent sets of three points), leaving no gap. Graphene behaves like a metal due to this property—and unlike superconductors, graphene can still conduct electricity at room temperature.
With that short science lecture finished, graphene’s remarkable conductive prowess has made it a hot-ticket material for some neurological applications. The interface between electrodes and neural tissues is essential to the recording and stimulation of neural signals. Due to an excellent signal-to-noise ratio (SNR), Graphene Flagship researchers recently used graphene-based transistors to test a flexible neural probe. The probe is able to record neurological activity in high resolution while maintaining an impressive SNR, revealing future possibilities for use in functional implants and interfaces.
The study entailed recording both large signals generated by pre-epileptic activity in rats, as well as lower levels of brain activity during sleep and response to visual light stimulation. Since neural activity is only detected through the localized electric fields when neurons fire, the ultra-small, densely-packed graphene measuring device is useful for accurate brain readings. Because the probes are placed directly on the brain, ensuring graphene’s safety in neural implants was also integral—the researchers determined the probes were non-toxic and didn’t induce significant inflammation.
Neural prostheses for therapeutic brain stimulation devices, as well as interfaces for sensory and motor devices are goals in improving quality of life that graphene may help to achieve. This work represents an initial step for high resolution and SNR in clinical graphene-based neural devices, but more research must be done.
“Graphene is one of the few materials that allows recording in a transistor configuration and simultaneously complies with all other requirements for neural probes such as flexibility, biocompability, and chemical stability,” commented Benno Blaschke, TU Munich researcher and first author of the study. “Although graphene is ideally suited for flexible electronics, it was a great challenge to transfer our fabrication process from rigid substrates to flexible ones. The next step is to optimize the wafer-scale fabrication process and improve device flexibility and stability.”
“Mechanical compliance is an important requirement for safe neural probes and interfaces,” noted Jose Antonio Garrido, Catalan Institute of Nanoscience and Nanotechnology researcher and lead researcher of the study. “Currently, the focus is on ultra-soft materials that can adapt conformally to the brain surface. Graphene neural interfaces have shown already great potential, but we have to improve on the yield and homogeneity of the device production in order to advance towards a real technology. Once we have demonstrated the proof of concept in animal studies, the next goal will be to work toward the first human clinical trial with graphene devices during intraoperative mapping of the brain. This means addressing all regulatory issues associated with medical devices such as safety, biocompatibility, etc.”
Researchers at the Daegu Gyeongbuk Institute of Science and Technology have investigated similar graphene-based applications, though with different methods. They too designed small, flexible graphene-based probes that clearly read neurological signals using tiny electrodes. However, this electrode begins with a thin gold base attached to zinc oxide nanowires coated in a thin gold layer and a conducting polymer called PEDOT [poly (3,4-ethylenedioxythiophene)]. Combined, the probe’s effective surface area, conducting properties, and electrode strength were increased while remaining flexible and compatible with soft tissue.
The smaller, graphene-based electrode retained the effective surface area of its larger counterpart due to several long, thin nanowires packed onto the probe. The electrode can then shrink without reduction in signal detection. The interconnection line—being composed of both graphene and gold—was both flexible and demonstrated excellent conductivity, outperforming a standard flat, gold electrode.
“Our graphene and nanowires-based flexible electrode array can be useful for monitoring and recording the functions of the nervous system, or to deliver electrical signals to the brain,” the researchers concluded in their paper recently published in ACS Applied Materials and Interfaces.
For a few years now, graphene has been tested for an impressive arsenal of futuristic medical technologies. It’s been researched as a platform to differentiate normal from cancerous cells, a replacement for titanium as a bone-filler, a nanoribbon to reconnect damaged spinal cords, and a strain sensor after insertion into silly putty.
Graphene-based electronics have the most promise in vaulting the material into the mainstream due to the its astounding electrical conductivity. Graphene assumes a flat, hexagonal lattice structure, which allows relatively low resistance to the flow of electrons. It fares better as a conductor than copper, reaching almost superconductor levels of power. Specifically, in its lattice form, graphene is known to be a “zero-gap semiconductor,” meaning its conduction and valence bonds meet at the Dirac points (six locations in momentum space on the edge of the Brillouin zone, divided into two non-equivalent sets of three points), leaving no gap. Graphene behaves like a metal due to this property—and unlike superconductors, graphene can still conduct electricity at room temperature.
With that short science lecture finished, graphene’s remarkable conductive prowess has made it a hot-ticket material for some neurological applications. The interface between electrodes and neural tissues is essential to the recording and stimulation of neural signals. Due to an excellent signal-to-noise ratio (SNR), Graphene Flagship researchers recently used graphene-based transistors to test a flexible neural probe. The probe is able to record neurological activity in high resolution while maintaining an impressive SNR, revealing future possibilities for use in functional implants and interfaces.
The study entailed recording both large signals generated by pre-epileptic activity in rats, as well as lower levels of brain activity during sleep and response to visual light stimulation. Since neural activity is only detected through the localized electric fields when neurons fire, the ultra-small, densely-packed graphene measuring device is useful for accurate brain readings. Because the probes are placed directly on the brain, ensuring graphene’s safety in neural implants was also integral—the researchers determined the probes were non-toxic and didn’t induce significant inflammation.
Neural prostheses for therapeutic brain stimulation devices, as well as interfaces for sensory and motor devices are goals in improving quality of life that graphene may help to achieve. This work represents an initial step for high resolution and SNR in clinical graphene-based neural devices, but more research must be done.
“Graphene is one of the few materials that allows recording in a transistor configuration and simultaneously complies with all other requirements for neural probes such as flexibility, biocompability, and chemical stability,” commented Benno Blaschke, TU Munich researcher and first author of the study. “Although graphene is ideally suited for flexible electronics, it was a great challenge to transfer our fabrication process from rigid substrates to flexible ones. The next step is to optimize the wafer-scale fabrication process and improve device flexibility and stability.”
“Mechanical compliance is an important requirement for safe neural probes and interfaces,” noted Jose Antonio Garrido, Catalan Institute of Nanoscience and Nanotechnology researcher and lead researcher of the study. “Currently, the focus is on ultra-soft materials that can adapt conformally to the brain surface. Graphene neural interfaces have shown already great potential, but we have to improve on the yield and homogeneity of the device production in order to advance towards a real technology. Once we have demonstrated the proof of concept in animal studies, the next goal will be to work toward the first human clinical trial with graphene devices during intraoperative mapping of the brain. This means addressing all regulatory issues associated with medical devices such as safety, biocompatibility, etc.”
Researchers at the Daegu Gyeongbuk Institute of Science and Technology have investigated similar graphene-based applications, though with different methods. They too designed small, flexible graphene-based probes that clearly read neurological signals using tiny electrodes. However, this electrode begins with a thin gold base attached to zinc oxide nanowires coated in a thin gold layer and a conducting polymer called PEDOT [poly (3,4-ethylenedioxythiophene)]. Combined, the probe’s effective surface area, conducting properties, and electrode strength were increased while remaining flexible and compatible with soft tissue.
The smaller, graphene-based electrode retained the effective surface area of its larger counterpart due to several long, thin nanowires packed onto the probe. The electrode can then shrink without reduction in signal detection. The interconnection line—being composed of both graphene and gold—was both flexible and demonstrated excellent conductivity, outperforming a standard flat, gold electrode.
“Our graphene and nanowires-based flexible electrode array can be useful for monitoring and recording the functions of the nervous system, or to deliver electrical signals to the brain,” the researchers concluded in their paper recently published in ACS Applied Materials and Interfaces.