02.06.15
Teleflex Medical OEM, a provider of precision extrusion and complex catheters, has added polyether ether ketone (PEEK) to its specialty materials portfolio.
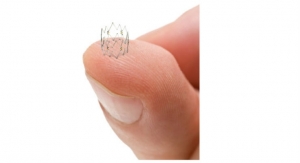
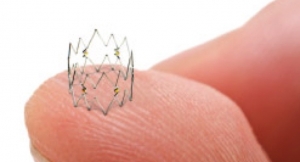
PEEK is known for its high tensile and column strength even though it is relatively lightweight. In fact, PEEK has one of the highest strength to weight ratios of any thermoplastic. The polymer often is used as a viable alternative for stainless steel and other metals in components for catheters used for coronary valve, stent, or graft delivery. In addition, PEEK has shown promise as a replacement for steel components used during many minimally invasive procedures. PEEK also demonstrates other high-performance characteristics such as dimensional stability, high burst pressure, chemical inertness, heat resistance, and outstanding lubricity.
A recent release from the company claims Teleflex OEM, the firm’s contract manufacturing division, can take a project from resin to tubing to finished device. It offers PEEK as a medical grade material that meets USP Class VI and cytotoxicity testing requirements. This material can be extruded as tubing with walls as thin as 0.005 inches while maintaining columnar stiffness, torsion strength, and low elongation. Custom options such as profiles, multi-lumen, and radiopaque fillers are available, officials said.
Teleflex makes specialty medical devices for a range of procedures in critical care and surgery. Based in Wayne, Pa., the company employs approximately 11,500 people worldwide and serves healthcare providers in more than 150 countries. Teleflex Medical OEM makes custom-engineered sutures and fibers, extrusions, diagnostic and interventional catheters, and sheath/dilator sets and kits. The company maintains product development and manufacturing facilities in the United States, Mexico and Ireland.
PEEK is known for its high tensile and column strength even though it is relatively lightweight. In fact, PEEK has one of the highest strength to weight ratios of any thermoplastic. The polymer often is used as a viable alternative for stainless steel and other metals in components for catheters used for coronary valve, stent, or graft delivery. In addition, PEEK has shown promise as a replacement for steel components used during many minimally invasive procedures. PEEK also demonstrates other high-performance characteristics such as dimensional stability, high burst pressure, chemical inertness, heat resistance, and outstanding lubricity.
A recent release from the company claims Teleflex OEM, the firm’s contract manufacturing division, can take a project from resin to tubing to finished device. It offers PEEK as a medical grade material that meets USP Class VI and cytotoxicity testing requirements. This material can be extruded as tubing with walls as thin as 0.005 inches while maintaining columnar stiffness, torsion strength, and low elongation. Custom options such as profiles, multi-lumen, and radiopaque fillers are available, officials said.
Teleflex makes specialty medical devices for a range of procedures in critical care and surgery. Based in Wayne, Pa., the company employs approximately 11,500 people worldwide and serves healthcare providers in more than 150 countries. Teleflex Medical OEM makes custom-engineered sutures and fibers, extrusions, diagnostic and interventional catheters, and sheath/dilator sets and kits. The company maintains product development and manufacturing facilities in the United States, Mexico and Ireland.