Christopher Delporte, Editorial Director02.06.15
One of the most dynamic aspects of being a medical device industry journalist for nearly 15 years is being able to observe the evolution of technology—as well as the companies behind the innovation. Nearly a year ago, Manchester, United Kingdom-based Zilico Ltd. introduced its primary product—the
. It was around that time I was introduced to the company and members of its leadership during an international industry trade show.
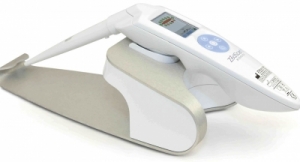
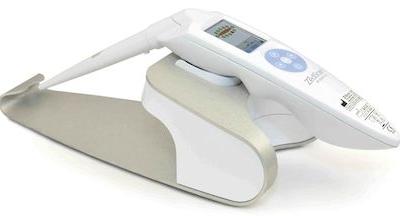
ZedScan is used in addition to colposcopy—the current standard of care—which involves a physician visually examining an illuminated, magnified view of the cervix and the tissues of the vagina and vulva. The company’s electrical impedance spectroscopy (EIS) technology helps to identify the different electrical resistivity of normal, pre-cancerous and cancerous tissue. The painless system consists of a portable handheld device and docking station, software application and a single-use EIS sensor.
So far, five clinical trials in the United Kingdom and the European Union have supported the technology’s efficacy, including a pivotal European trial in 2011 of 429 women in three hospitals in the United Kingdom and Ireland.
ZedScan is CE marked, and the company recently signed a three-year deal with a U.K. distributor.
A lot can happen in a year.
“ZedScan has generated substantial interest from distributors across the globe through various presentations, conferences and clinical meetings,” said Sameer Kothari, Zilico’s chief executive. “Now we look forward to being able to satisfy the growing demand starting at the world’s biggest healthcare exhibition.”
Kothari was on hand during last fall’s Medica 2014 held in Düsseldorf, Germany, to demonstrate and discuss his company’s technology as well as plans for new products based on EIS.
The company is pursuing U.S. Food and Drug Administration approval, and is in preliminary discussions with the agency, Kothari confirmed to Medical Product Outsourcing. In the meantime, the Zilico will continue to gather more data with evaluation studies in Australia, France, Germany, Canada, Sweden and Israel.
“Clinicians like the fact this [technology] fits into their practice,” Kothari said. “They don’t like to change things.”
Zilico is working on a medical device that uses EIS to help detect oral cancer, Kothari said. Oral cancer can be difficult to diagnose.
“About 90 percent of biopsy samples for oral cancer come back negative,” Kothari told MPO. “There’s a need for real-time info to help determine if a lesion in the mouth might be malignant or not. This is very valuable.”
The company has started with a small 50-person proof-of-concept study that recently was chronicled in the Journal of Nanotechnology.
The study aimed to determine whether electrical impedance spectroscopy could distinguish between normal oral mucosa; benign; potentially malignant lesions (PML) and oral cancer. The data was presented during the 67th annual meeting of the American Academy of Oral Medicine in San Antonio, Texas.
“Study subjects also deemed the EIS procedure considerably less painful and more convenient than the scalpel biopsy procedure. EIS shows promise at distinguishing among malignant, PML, and normal oral mucosa and has the potential to be developed into a clinical diagnostic tool,” the Journal article concluded.
The technology for the oral cancer product and ZedScan would be virtually identical, though the handheld device would need to be specially reconfigured for use in the mouth. Oral cancer follows a similar neoplastic pathway to that of the cervix, so EIS can be used to detect changes in the oral epithelium as cells progress from normal to precancerous and then to cancerous.
The decision to move forward with the oral cancer product was made after the U.K.’s innovation agency, the Technology Strategy Board, agreed to fund the project.
The two awards are from the Biomedical Catalyst Feasibility and Smart Funds. The funds will help support an in-depth analysis of the U.K. market, deliver a product specification for an oral diagnostic device and a protocol for a multi-center trial. The awards will help support development projects worth approximately $250,000.
Following oral cancer, the technology also could be applied to anal and esophageal cancers as well, Kothari said, adding that the company also is evaluating a “slightly different” product designed for intraoperative use.
Oh, and in case you were wondering about the company’s name:
Z1ilico2
ZedScan is used in addition to colposcopy—the current standard of care—which involves a physician visually examining an illuminated, magnified view of the cervix and the tissues of the vagina and vulva. The company’s electrical impedance spectroscopy (EIS) technology helps to identify the different electrical resistivity of normal, pre-cancerous and cancerous tissue. The painless system consists of a portable handheld device and docking station, software application and a single-use EIS sensor.
So far, five clinical trials in the United Kingdom and the European Union have supported the technology’s efficacy, including a pivotal European trial in 2011 of 429 women in three hospitals in the United Kingdom and Ireland.
ZedScan is CE marked, and the company recently signed a three-year deal with a U.K. distributor.
A lot can happen in a year.
“ZedScan has generated substantial interest from distributors across the globe through various presentations, conferences and clinical meetings,” said Sameer Kothari, Zilico’s chief executive. “Now we look forward to being able to satisfy the growing demand starting at the world’s biggest healthcare exhibition.”
Kothari was on hand during last fall’s Medica 2014 held in Düsseldorf, Germany, to demonstrate and discuss his company’s technology as well as plans for new products based on EIS.
The company is pursuing U.S. Food and Drug Administration approval, and is in preliminary discussions with the agency, Kothari confirmed to Medical Product Outsourcing. In the meantime, the Zilico will continue to gather more data with evaluation studies in Australia, France, Germany, Canada, Sweden and Israel.
“Clinicians like the fact this [technology] fits into their practice,” Kothari said. “They don’t like to change things.”
Zilico is working on a medical device that uses EIS to help detect oral cancer, Kothari said. Oral cancer can be difficult to diagnose.
“About 90 percent of biopsy samples for oral cancer come back negative,” Kothari told MPO. “There’s a need for real-time info to help determine if a lesion in the mouth might be malignant or not. This is very valuable.”
The company has started with a small 50-person proof-of-concept study that recently was chronicled in the Journal of Nanotechnology.
The study aimed to determine whether electrical impedance spectroscopy could distinguish between normal oral mucosa; benign; potentially malignant lesions (PML) and oral cancer. The data was presented during the 67th annual meeting of the American Academy of Oral Medicine in San Antonio, Texas.
“Study subjects also deemed the EIS procedure considerably less painful and more convenient than the scalpel biopsy procedure. EIS shows promise at distinguishing among malignant, PML, and normal oral mucosa and has the potential to be developed into a clinical diagnostic tool,” the Journal article concluded.
The technology for the oral cancer product and ZedScan would be virtually identical, though the handheld device would need to be specially reconfigured for use in the mouth. Oral cancer follows a similar neoplastic pathway to that of the cervix, so EIS can be used to detect changes in the oral epithelium as cells progress from normal to precancerous and then to cancerous.
The decision to move forward with the oral cancer product was made after the U.K.’s innovation agency, the Technology Strategy Board, agreed to fund the project.
The two awards are from the Biomedical Catalyst Feasibility and Smart Funds. The funds will help support an in-depth analysis of the U.K. market, deliver a product specification for an oral diagnostic device and a protocol for a multi-center trial. The awards will help support development projects worth approximately $250,000.
Following oral cancer, the technology also could be applied to anal and esophageal cancers as well, Kothari said, adding that the company also is evaluating a “slightly different” product designed for intraoperative use.
Oh, and in case you were wondering about the company’s name:
Z1ilico2
- Z (zeta) is the symbol for impedance, the all-important measure that company leadership says underpins Zilico’s technology.
- ilico is a Latin word meaning “on the spot” or “immediate”—reflecting the benefit of diagnostic devices that deliver results in real time.