06.14.13
Sunnyvale, Calif.-based Elixir Medical Corp. has been granted investigational device exemption (IDE) status approval from the U.S. Food and Drug Administration (FDA) to initiate patient enrollment in the Excella III clinical trial in the United States at up to 50 institutions. Results from the Excella III pivotal trial will be used to support a future pre-market approval (PMA) application in the United States for the Desyne Nx Novolimus-eluting coronary stent system.
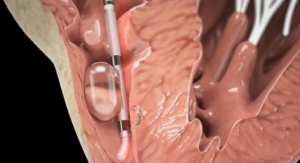
The Desyne Nx stent is designed to treat heart vessel blockages by eluting a low dose of Elixir’s proprietary drug Novolimus, an active metabolite of sirolimus (an immunosuppressant), via an ultra-thin durable coating. The Excella III clinical trial will be a prospective, controlled, multicenter, single-blind study comparing Desyne Nx to Medtronic Inc.’s Resolute Zotarolimus-eluting coronary stent system as a control in a 2:1 randomization of 2,051 patients recruited from U.S. and international centers. The principal investigator is Martin B. Leon, M.D., of New York–Presbyterian Hospital and Columbia University Medical Center, New York, N.Y.
The IDE approval for the Excella III trial in the United States follows the international commercial launch of Desyne, which gained CE mark approval based on the results of the Excella II trial. The Excella II trial was a randomized controlled study that enrolled 210 patients at 22 clinical centers in Europe, Australia, New Zealand and Brazil. The three-year follow-up results were presented in October 2012 at the Transcatheter Therapeutics Conference in Washington, D.C. The Desyne stent reportedly demonstrated both non-inferiority and superiority to the Zotarolimus-eluting control stent for the primary endpoint of in-stent late lumen loss and achieved low clinical event rate through three years. The data collected from the global use of Desyne also will be used to supplement a future PMA application in the United States for the Desyne Nx Novolimus eluting coronary stent system.
“Excella III will be a promising trial building upon the excellent and sustained clinical outcomes of the Excella II trial,” said Leon. “I am excited about Desyne Nx’s potential to validate the safety and long-term performance in this large clinical trial.”
The primary endpoint of the Excella III trial is target lesion failure, a composite measure of safety and effectiveness at 12 months defined as cardiac death, myocardial infarction related to the target vessel, and clinically-indicated target lesion revascularization. In addition, a subset of patients will be evaluated for angiographic endpoints, and all patients will be followed for five years.
“We are excited to bring into the United States the next-generation DES that successfully combines the thinnest durable polymer coating with the lowest drug dose and thin stent struts for clinical evaluation,” said Motasim Sirhan, CEO of Elixir Medical. “Elixir’s goal is to bring its excellent and comprehensive product portfolio to patients globally, and the IDE approval for Desyne Nx is an important milestone towards this objective.”
The Desyne Nx stent is designed to treat heart vessel blockages by eluting a low dose of Elixir’s proprietary drug Novolimus, an active metabolite of sirolimus (an immunosuppressant), via an ultra-thin durable coating. The Excella III clinical trial will be a prospective, controlled, multicenter, single-blind study comparing Desyne Nx to Medtronic Inc.’s Resolute Zotarolimus-eluting coronary stent system as a control in a 2:1 randomization of 2,051 patients recruited from U.S. and international centers. The principal investigator is Martin B. Leon, M.D., of New York–Presbyterian Hospital and Columbia University Medical Center, New York, N.Y.
The IDE approval for the Excella III trial in the United States follows the international commercial launch of Desyne, which gained CE mark approval based on the results of the Excella II trial. The Excella II trial was a randomized controlled study that enrolled 210 patients at 22 clinical centers in Europe, Australia, New Zealand and Brazil. The three-year follow-up results were presented in October 2012 at the Transcatheter Therapeutics Conference in Washington, D.C. The Desyne stent reportedly demonstrated both non-inferiority and superiority to the Zotarolimus-eluting control stent for the primary endpoint of in-stent late lumen loss and achieved low clinical event rate through three years. The data collected from the global use of Desyne also will be used to supplement a future PMA application in the United States for the Desyne Nx Novolimus eluting coronary stent system.
“Excella III will be a promising trial building upon the excellent and sustained clinical outcomes of the Excella II trial,” said Leon. “I am excited about Desyne Nx’s potential to validate the safety and long-term performance in this large clinical trial.”
The primary endpoint of the Excella III trial is target lesion failure, a composite measure of safety and effectiveness at 12 months defined as cardiac death, myocardial infarction related to the target vessel, and clinically-indicated target lesion revascularization. In addition, a subset of patients will be evaluated for angiographic endpoints, and all patients will be followed for five years.
“We are excited to bring into the United States the next-generation DES that successfully combines the thinnest durable polymer coating with the lowest drug dose and thin stent struts for clinical evaluation,” said Motasim Sirhan, CEO of Elixir Medical. “Elixir’s goal is to bring its excellent and comprehensive product portfolio to patients globally, and the IDE approval for Desyne Nx is an important milestone towards this objective.”