07.19.16
$5.5 Billion
KEY EXECUTIVES:
Michael T. Rousseau, President and CEO
John C. Heinmiller, Exec. VP President, Americas Division
Mark D. Carlson, M.D., VP, Global Clinical Affairs and Chief Medical Officer
Philip J. Ebeling, VP, Chief Technology Officer
Jeffry A. Fecho, VP, Global Quality
Denis M. Gestin, President, International Division
Scott P. Thome, VP, Global Operations and Supply Chain
Jason A. Zellers, VP, General Counsel and Corporate Secretary
Donald J. Zurbay, VP, Finance and Chief Financial Officer
NUMBER OF EMPLOYEES: 18,000
GLOBAL HEADQUARTERS: St. Paul, Minn.
Last year saw St. Jude Medical Inc.’s largest acquisition yet. In October, the company bought Thoratec Corporation, which makes mechanical circulatory support technology for the treatment of advanced heart failure (HF).
The acquisition added the HeartMate II ventricular assist device, as well as the next-generation HeartMate 3, HeartMate PHP devices, and other complementary products to St. Jude Medical’s heart failure portfolio. With the addition of these product lines, St. Jude Medical now offers a significantly enhanced portfolio of devices for the management and treatment of heart failure.
The deal was valued at approximately $3.3 billion, net of cash acquired. Under the terms of the transaction, each outstanding share of Thoratec at the time of the closing was converted into the right to receive $63.50 in cash, without interest. St. Jude Medical expects the acquisition to be accretive to adjusted earnings per share in 2016.
Zacks analysts noted that St. Jude Medical is not accustomed to large takeovers. The analyst firm predicted the integration may cost the company more than it bargained for in 2016. However, this potential problem resolved itself earlier this year when Abbott one-upped the St. Jude-Thoratec deal by purchasing St. Jude Medical itself for $25 billion.
Financial Results
For the full-year 2015 (ended Dec. 31), net sales were $5.54 billion, compared with $5.62 billion in 2014. On a constant-currency basis, net sales increased 6 percent over the prior year. When comparing net sales for the full year 2015 to the combined net sales of both St. Jude Medical and Thoratec for the full-year 2014 and adjusting for foreign currency, comparable constant currency sales increased 4 percent.
Commenting on the company’s financial results, St. Jude Medical President and Chief Executive Officer Michael T. Rousseau said, “We are pleased with the momentum we achieved in 2015 in our atrial fibrillation, heart failure, and neuromodulation businesses, and with the significant progress we have made integrating Thoratec, which had a strong fourth quarter. In 2016, we expect to continue our focus on these key areas which will drive our growth and allow us to deliver a comprehensive portfolio of innovative technologies to patients around the world.”
St. Jude Medical operates on a 52/53 week fiscal year convention with an extra 53rd week occurring approximately every six years. The company’s fiscal fourth quarter 2014 had one extra week of sales compared to fiscal fourth quarter 2015. The company’s fiscal year 2014 had three extra selling days compared to fiscal year 2015. In order to provide similar sales comparisons for quarterly and annual results, the company recommends adjusting for the impact of this convention. While the calculation is not precise, the company estimates that these fewer selling days in 2015 negatively impacted fourth quarter 2015 results by approximately 5 to 6 percentage points and full-year 2015 results by approximately 1 percentage point.
Atrial Fibrillation Sales
For the full-year 2015, Atrial Firbrillation (AF) product sales were $1.09 billion, an increase of 5 percent over the prior year. On a constant-currency basis, AF product sales increased 13 percent in 2015.
St. Jude Medical significantly improved its ablation catheter offerings in its cardiac arrhythmia product portfolio last year with the commercialization of two new catheters. The TactiCath Quartz contact-force sensing irrigated ablation catheter pioneered the ability to give physicians a realtime, objective measure of the force applied to the heart wall during a cardiac ablation procedure to treat paroxysmal AF. The company’s FlexAbility ablation catheter was designed with feedback from leaders in the global electrophysiology community to enhance the quality of care for patients during ablation procedures. According to St. Jude Medical, the product introductions have allowed physicians to tailor patient therapy for optimized outcomes and position the company as the provider of choice to electrophysiology customers.
Cardiovascular and Cardiac Rhythm Management (CRM) Performance
CRM sales last year totaled $2.52 billion, a 10 percent decrease when compared with 2014. On a constant-currency basis, total CRM sales decreased 3 percent from the prior year.
Total cardiovascular product sales for 2015 were $1.31 billion, a decrease of 3 percent from the prior year. On a constant-currency basis, cardiovascular product sales increased 6 percent in 2015.
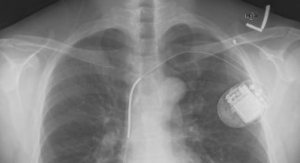
The CardioMEMS HF System is the cornerstone of St. Jude Medical’s heart failure product portfolio. The company claims it is the only firm with a U.S. Food and Drug Administration (FDA)-approved remote hemodynamic monitoring device.
In the November 2015 edition of The Lancet, prospective data from the CHAMPION study showed that after a mean of 31 months of follow-up, HF patients managed with the CardioMEMS HF system had a 48 percent reduction in HF hospitalizations compared to patients managed with the current standard of care. The data confirms the benefits of the technology and has driven strong clinical acceptance. However, with new innovative technologies, reimbursement can lag behind regulatory approval. Thus, St. Jude Medical is pursuing a National Coverage Determination from the Centers for Medicare & Medicaid Services to ensure that indicated patients have access to the CardioMEMS HF system. The process is expected to be completed by the end of this year and the company continues to develop this new market by expanding clinical evidence to drive adoption and reimbursement globally. St. Jude Medical’s acquisition of Thoratec—the largest in the company’s history—positions the company as the market leader in left ventricular assist devices (LVADs).
In 2015 St. Jude Medical announced CE mark for the HeartMate 3 Left Ventricular Assist System, a significant new product for its HF portfolio. The company expects the LVAD market to continue to be an attractive growth market in 2016, and expects to continue developing the market, since currently only about 10 percent of eligible patients receive this life-saving therapy. The HeartMate 3 LVAS international launch has gone well, and the HeartMate 3 LVAS U.S. investigational device exemption clinical study, MOMENTUM, is making good progress with patient enrollment.
The company also received CE mark approval in December last year to add magnetic resonance conditional labeling for its Quadra Assura CRT-D and Quadra Assura MP CRT-D with MultiPoint Pacing technology.
Neuromodulation Revenue
Neuromodulation product sales in 2015 were $475 million, an increase of 9 percent over the prior year. On a constant currency basis, however, Neuromodulation product sales increased 14 percent last year.
By adding Spinal Modulation Inc., and its Axium Neurostimulator System (approved by the FDA in February 2016) to its offerings in 2015, St. Jude Medical is now the only medical device manufacturer to offer radiofrequency ablation, spinal cord stimulation (SCS), and dorsal root ganglion stimulation therapy solutions for the treatment of chronic pain. These two acquisitions, supported by compelling clinical data, offer unique proprietary solutions that change the landscape of the chronic pain management market.
In 2015, the St. Jude Medical Invisible Trial System received approval in the United States and Europe. The system is fully wireless and leverages Apple technology for both the patient and physician controllers. Developed with physician and patient feedback, the system is designed to offer patients an improved and discreet SCS trial experience. Also approved in Europe in 2015, the Proclaim Elite Spinal Cord Stimulation System is the first and only upgradeable and non-rechargeable SCS system approved to deliver Burst stimulation.
Also in 2015, SUNBURST trial results analyzing St. Jude Medical’s Burst technology demonstrated a reduction or elimination of paresthesia in 91 percent of patients. The company plans to establish Burst therapy as the dominant spinal cord stimulation waveform to achieve superior pain relief across rechargeable and rechargeable-free devices.
KEY EXECUTIVES:
Michael T. Rousseau, President and CEO
John C. Heinmiller, Exec. VP President, Americas Division
Mark D. Carlson, M.D., VP, Global Clinical Affairs and Chief Medical Officer
Philip J. Ebeling, VP, Chief Technology Officer
Jeffry A. Fecho, VP, Global Quality
Denis M. Gestin, President, International Division
Scott P. Thome, VP, Global Operations and Supply Chain
Jason A. Zellers, VP, General Counsel and Corporate Secretary
Donald J. Zurbay, VP, Finance and Chief Financial Officer
NUMBER OF EMPLOYEES: 18,000
GLOBAL HEADQUARTERS: St. Paul, Minn.
Last year saw St. Jude Medical Inc.’s largest acquisition yet. In October, the company bought Thoratec Corporation, which makes mechanical circulatory support technology for the treatment of advanced heart failure (HF).
The acquisition added the HeartMate II ventricular assist device, as well as the next-generation HeartMate 3, HeartMate PHP devices, and other complementary products to St. Jude Medical’s heart failure portfolio. With the addition of these product lines, St. Jude Medical now offers a significantly enhanced portfolio of devices for the management and treatment of heart failure.
The deal was valued at approximately $3.3 billion, net of cash acquired. Under the terms of the transaction, each outstanding share of Thoratec at the time of the closing was converted into the right to receive $63.50 in cash, without interest. St. Jude Medical expects the acquisition to be accretive to adjusted earnings per share in 2016.
Zacks analysts noted that St. Jude Medical is not accustomed to large takeovers. The analyst firm predicted the integration may cost the company more than it bargained for in 2016. However, this potential problem resolved itself earlier this year when Abbott one-upped the St. Jude-Thoratec deal by purchasing St. Jude Medical itself for $25 billion.
Financial Results
For the full-year 2015 (ended Dec. 31), net sales were $5.54 billion, compared with $5.62 billion in 2014. On a constant-currency basis, net sales increased 6 percent over the prior year. When comparing net sales for the full year 2015 to the combined net sales of both St. Jude Medical and Thoratec for the full-year 2014 and adjusting for foreign currency, comparable constant currency sales increased 4 percent.
Commenting on the company’s financial results, St. Jude Medical President and Chief Executive Officer Michael T. Rousseau said, “We are pleased with the momentum we achieved in 2015 in our atrial fibrillation, heart failure, and neuromodulation businesses, and with the significant progress we have made integrating Thoratec, which had a strong fourth quarter. In 2016, we expect to continue our focus on these key areas which will drive our growth and allow us to deliver a comprehensive portfolio of innovative technologies to patients around the world.”
St. Jude Medical operates on a 52/53 week fiscal year convention with an extra 53rd week occurring approximately every six years. The company’s fiscal fourth quarter 2014 had one extra week of sales compared to fiscal fourth quarter 2015. The company’s fiscal year 2014 had three extra selling days compared to fiscal year 2015. In order to provide similar sales comparisons for quarterly and annual results, the company recommends adjusting for the impact of this convention. While the calculation is not precise, the company estimates that these fewer selling days in 2015 negatively impacted fourth quarter 2015 results by approximately 5 to 6 percentage points and full-year 2015 results by approximately 1 percentage point.
Atrial Fibrillation Sales
For the full-year 2015, Atrial Firbrillation (AF) product sales were $1.09 billion, an increase of 5 percent over the prior year. On a constant-currency basis, AF product sales increased 13 percent in 2015.
St. Jude Medical significantly improved its ablation catheter offerings in its cardiac arrhythmia product portfolio last year with the commercialization of two new catheters. The TactiCath Quartz contact-force sensing irrigated ablation catheter pioneered the ability to give physicians a realtime, objective measure of the force applied to the heart wall during a cardiac ablation procedure to treat paroxysmal AF. The company’s FlexAbility ablation catheter was designed with feedback from leaders in the global electrophysiology community to enhance the quality of care for patients during ablation procedures. According to St. Jude Medical, the product introductions have allowed physicians to tailor patient therapy for optimized outcomes and position the company as the provider of choice to electrophysiology customers.
Cardiovascular and Cardiac Rhythm Management (CRM) Performance
CRM sales last year totaled $2.52 billion, a 10 percent decrease when compared with 2014. On a constant-currency basis, total CRM sales decreased 3 percent from the prior year.
Total cardiovascular product sales for 2015 were $1.31 billion, a decrease of 3 percent from the prior year. On a constant-currency basis, cardiovascular product sales increased 6 percent in 2015.
The CardioMEMS HF System is the cornerstone of St. Jude Medical’s heart failure product portfolio. The company claims it is the only firm with a U.S. Food and Drug Administration (FDA)-approved remote hemodynamic monitoring device.
In the November 2015 edition of The Lancet, prospective data from the CHAMPION study showed that after a mean of 31 months of follow-up, HF patients managed with the CardioMEMS HF system had a 48 percent reduction in HF hospitalizations compared to patients managed with the current standard of care. The data confirms the benefits of the technology and has driven strong clinical acceptance. However, with new innovative technologies, reimbursement can lag behind regulatory approval. Thus, St. Jude Medical is pursuing a National Coverage Determination from the Centers for Medicare & Medicaid Services to ensure that indicated patients have access to the CardioMEMS HF system. The process is expected to be completed by the end of this year and the company continues to develop this new market by expanding clinical evidence to drive adoption and reimbursement globally. St. Jude Medical’s acquisition of Thoratec—the largest in the company’s history—positions the company as the market leader in left ventricular assist devices (LVADs).
In 2015 St. Jude Medical announced CE mark for the HeartMate 3 Left Ventricular Assist System, a significant new product for its HF portfolio. The company expects the LVAD market to continue to be an attractive growth market in 2016, and expects to continue developing the market, since currently only about 10 percent of eligible patients receive this life-saving therapy. The HeartMate 3 LVAS international launch has gone well, and the HeartMate 3 LVAS U.S. investigational device exemption clinical study, MOMENTUM, is making good progress with patient enrollment.
The company also received CE mark approval in December last year to add magnetic resonance conditional labeling for its Quadra Assura CRT-D and Quadra Assura MP CRT-D with MultiPoint Pacing technology.
Neuromodulation Revenue
Neuromodulation product sales in 2015 were $475 million, an increase of 9 percent over the prior year. On a constant currency basis, however, Neuromodulation product sales increased 14 percent last year.
By adding Spinal Modulation Inc., and its Axium Neurostimulator System (approved by the FDA in February 2016) to its offerings in 2015, St. Jude Medical is now the only medical device manufacturer to offer radiofrequency ablation, spinal cord stimulation (SCS), and dorsal root ganglion stimulation therapy solutions for the treatment of chronic pain. These two acquisitions, supported by compelling clinical data, offer unique proprietary solutions that change the landscape of the chronic pain management market.
In 2015, the St. Jude Medical Invisible Trial System received approval in the United States and Europe. The system is fully wireless and leverages Apple technology for both the patient and physician controllers. Developed with physician and patient feedback, the system is designed to offer patients an improved and discreet SCS trial experience. Also approved in Europe in 2015, the Proclaim Elite Spinal Cord Stimulation System is the first and only upgradeable and non-rechargeable SCS system approved to deliver Burst stimulation.
Also in 2015, SUNBURST trial results analyzing St. Jude Medical’s Burst technology demonstrated a reduction or elimination of paresthesia in 91 percent of patients. The company plans to establish Burst therapy as the dominant spinal cord stimulation waveform to achieve superior pain relief across rechargeable and rechargeable-free devices.