Bill Stearns, Associate Senior Engineer, Intertek04.17.20
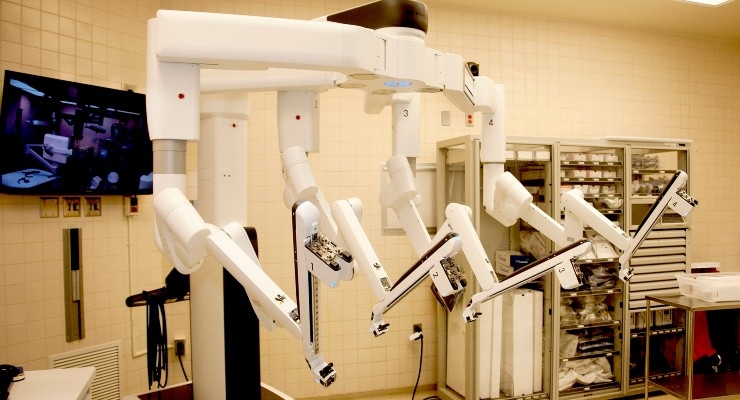
The use of robotic devices in medical applications and settings started years ago, with robotics assisting in surgical procedures. Since then, it has evolved to include more applications in surgery, as well as rehabilitation, assessment, compensation, or alleviation related to the patient’s movement functions. This evolution means rapid, widespread growth in the industry. It is estimated the global market for surgical robots will nearly triple in a decade, from $4.5 billion in 2016 to $12.6 billion in 2025.1
As robots and robotics become more prevalent in everyday life, it is understandable the medical industry seeks to capitalize on medical robots for their various applications and benefits. As manufacturers look to bring these innovative devices to market, it is important to be aware of potential hazards robots may present and testing options to address them. It is also critical to know and understand the multiple standards and technical reports that apply to these products.
Applicable Standards for the Evaluation of Medical Robots
Medical robots, like other medical equipment, must meet IEC 60601-1 requirements for basic safety and essential performance. Some of the basic safety requirements include prevention of shock, fire, excessive temperatures, radiation exposure, mechanical hazards, and electromagnetic emissions. Basic safety must be maintained in normal and single fault conditions. The standard also requires a robot to meet essential performance requirements defined by the manufacturer’s risk assessment.
Additional requirements are defined in the recently released standards IEC 80601-2-77 for Robotically Assisted Surgical Equipment and IEC 80601-2-78 for Medical Robots for rehabilitation, assessment compensation, or alleviation. Information essential to perform surgery, motion control, or force/torque of movement are some examples of essential performance. The manufacturer’s risk management must address all aspects that can contribute to the loss of essential performance, including breakdown of hardware, software, and hazards relating to usability—including system misuse. The loss of essential performance that could lead to patient harm must be minimized and a risk-benefit analysis must be performed for all hazards that cannot be reduced to an acceptable level.
IEC 60601-1 has two sections to consider regarding medical robots:
IEC 60601 1:2005 +A1:2012 General Requirements for Basic Safety and Essential Performance: Focuses on medical electrical equipment safety and essential performance. It is used throughout the world, including in the U.S., where it can be used toward U.S. Food and Drug Administration (FDA) approval. The standard—along with its many collateral and particular standards—includes requirements and considerations for risk assessment, software, usability, EMC, essential performance, and basic safety:
IEC 60601-1-11 Home healthcare: Meeting these requirements will be necessary if the robot is used in a home healthcare environment. Hazards related to controls, the environment, and misuse must be considered. Additional testing is performed to address some of these hazards and the user manual and markings need to be provided at a level that non-professional users can understand.
IEC 80601-2 also applies to medical robots, with two sections to consider for robotic devices:
IEC 80601-2-77: Specific to robotic-assisted surgical equipment and systems. This standard includes requirements for basic safety and essential performance of these robots. Additional requirements related to markings, warnings, safety notices, and protection against mechanical hazards are detailed. Risk controls for motion including: continuous activation, movement in the view of the operator, end stops, and different levels of protective stop functions are included. Essential performance and requirements relating to patient release, collisions, and compatibility with other medical systems are provided. It also recognizes robotic equipment is complex and provides mechanisms to reduce spacing in certain situations to allow for very dense electronic designs.
IEC 80601-2-78: Specific to robots used for rehabilitation, assessment, compensation, or alleviation related to the patient’s movement. It covers basic safety and essential performance. It applies to medical robots that physically interact with an impaired patient to support movement functions. The standard defines additional requirements for essential performance, protective sopping procedures, unintended movement, emergency stopping devices, release of patient, and mechanical hazards related to support systems. It also provides additional requirements for usability and specific test requirements.
Navigating these standards, documentation requirements, and various assessments may prove daunting at first glance. However, many of the standards address the hazards and assessments to be cognizant of. Educating yourself about the standards, hazards, and testing options will help design and produce a robotic device that is as safe and high-performing as possible. As the field continues to evolve, it is also important to stay updated on changes, additions, or introduction of new standards. Working with knowledgeable testing partners familiar with requisite standards can also be beneficial. They will be aware of industry developments and changes and work as a part of your team.
Innovation in robotic technology and an ever-changing healthcare landscape are coming together to pave the way for medical robots and robotic devices to become more prevalent. As standards emerge and evolve to ensure safety and performance of these cutting-edge products, be aware of the requirements to ensure compliance and bring compliant devices to market.
Reference
1 Statista. Global market size for surgical robots in 2016, 2017, and a forecast for 2025 (in billion U.S. dollars). https://www.statista.com/statistics/877720/surgical-robots-market-value-worldwide/. Accessed 3/31/2020
Bill Stearns is an associate senior engineer at Intertek, a Total Quality Assurance provider to industries worldwide.
As robots and robotics become more prevalent in everyday life, it is understandable the medical industry seeks to capitalize on medical robots for their various applications and benefits. As manufacturers look to bring these innovative devices to market, it is important to be aware of potential hazards robots may present and testing options to address them. It is also critical to know and understand the multiple standards and technical reports that apply to these products.
Applicable Standards for the Evaluation of Medical Robots
Medical robots, like other medical equipment, must meet IEC 60601-1 requirements for basic safety and essential performance. Some of the basic safety requirements include prevention of shock, fire, excessive temperatures, radiation exposure, mechanical hazards, and electromagnetic emissions. Basic safety must be maintained in normal and single fault conditions. The standard also requires a robot to meet essential performance requirements defined by the manufacturer’s risk assessment.
Additional requirements are defined in the recently released standards IEC 80601-2-77 for Robotically Assisted Surgical Equipment and IEC 80601-2-78 for Medical Robots for rehabilitation, assessment compensation, or alleviation. Information essential to perform surgery, motion control, or force/torque of movement are some examples of essential performance. The manufacturer’s risk management must address all aspects that can contribute to the loss of essential performance, including breakdown of hardware, software, and hazards relating to usability—including system misuse. The loss of essential performance that could lead to patient harm must be minimized and a risk-benefit analysis must be performed for all hazards that cannot be reduced to an acceptable level.
IEC 60601-1 has two sections to consider regarding medical robots:
IEC 60601 1:2005 +A1:2012 General Requirements for Basic Safety and Essential Performance: Focuses on medical electrical equipment safety and essential performance. It is used throughout the world, including in the U.S., where it can be used toward U.S. Food and Drug Administration (FDA) approval. The standard—along with its many collateral and particular standards—includes requirements and considerations for risk assessment, software, usability, EMC, essential performance, and basic safety:
- Risk management: A risk management evaluation complying with ISO 14971 must be performed. This standard provides the processes to evaluate hazards and reduce them to acceptable levels. The process includes the following steps: determine the policy for risk acceptability, identify hazards, estimation of risk, risk evaluation, risk control options, implementation of risk control measures, verification of risk control, and overall risk acceptability. All aspects of the design are part of this evaluation.
- Programmable medical systems: Evaluated according to clause 14 and IEC 62304. Hazards related to software must be included in the risk assessment according to the ISO 14971 process.
- Usability: Usability of the medical equipment is evaluated according to IEC 60601-1-6 and IEC 62366. A newer version (IEC 62366-1) is also required in many cases. The manufacturer should consider both standards when performing the usability evaluations. Hazards relating to usability must also be included in the risk management process.
IEC 60601-1-11 Home healthcare: Meeting these requirements will be necessary if the robot is used in a home healthcare environment. Hazards related to controls, the environment, and misuse must be considered. Additional testing is performed to address some of these hazards and the user manual and markings need to be provided at a level that non-professional users can understand.
IEC 80601-2 also applies to medical robots, with two sections to consider for robotic devices:
IEC 80601-2-77: Specific to robotic-assisted surgical equipment and systems. This standard includes requirements for basic safety and essential performance of these robots. Additional requirements related to markings, warnings, safety notices, and protection against mechanical hazards are detailed. Risk controls for motion including: continuous activation, movement in the view of the operator, end stops, and different levels of protective stop functions are included. Essential performance and requirements relating to patient release, collisions, and compatibility with other medical systems are provided. It also recognizes robotic equipment is complex and provides mechanisms to reduce spacing in certain situations to allow for very dense electronic designs.
IEC 80601-2-78: Specific to robots used for rehabilitation, assessment, compensation, or alleviation related to the patient’s movement. It covers basic safety and essential performance. It applies to medical robots that physically interact with an impaired patient to support movement functions. The standard defines additional requirements for essential performance, protective sopping procedures, unintended movement, emergency stopping devices, release of patient, and mechanical hazards related to support systems. It also provides additional requirements for usability and specific test requirements.
Navigating these standards, documentation requirements, and various assessments may prove daunting at first glance. However, many of the standards address the hazards and assessments to be cognizant of. Educating yourself about the standards, hazards, and testing options will help design and produce a robotic device that is as safe and high-performing as possible. As the field continues to evolve, it is also important to stay updated on changes, additions, or introduction of new standards. Working with knowledgeable testing partners familiar with requisite standards can also be beneficial. They will be aware of industry developments and changes and work as a part of your team.
Innovation in robotic technology and an ever-changing healthcare landscape are coming together to pave the way for medical robots and robotic devices to become more prevalent. As standards emerge and evolve to ensure safety and performance of these cutting-edge products, be aware of the requirements to ensure compliance and bring compliant devices to market.
Reference
1 Statista. Global market size for surgical robots in 2016, 2017, and a forecast for 2025 (in billion U.S. dollars). https://www.statista.com/statistics/877720/surgical-robots-market-value-worldwide/. Accessed 3/31/2020
Bill Stearns is an associate senior engineer at Intertek, a Total Quality Assurance provider to industries worldwide.