Sam Brusco, Associate Editor02.28.24
Brooklyn Hall is a first-grader who enjoys spending time on the tennis court. She has a diagnosis of Type 1 diabetes—an enormous burden for anyone, let alone a child. She has to both pay close attention to her glucose numbers and take regular doses of insulin.
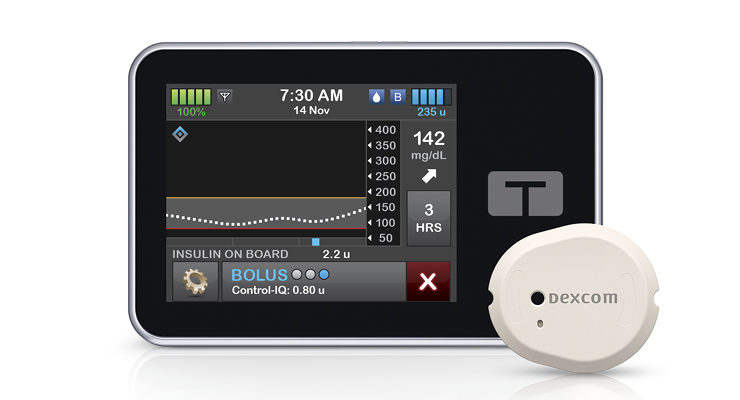
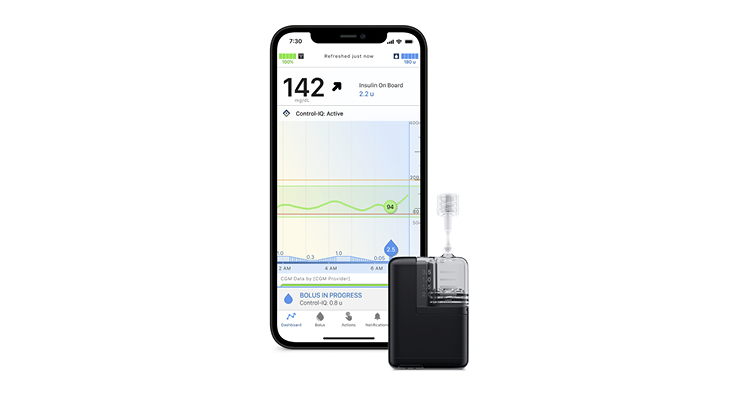
Thankfully, modern medicine has the technology to help young Brooklyn manage her illness so she can spend more time doing the things she loves—and less time worrying about glucose numbers and insulin injections. She wears both the Tandem t:slim X2 insulin pump and Dexcom G7 continuous glucose monitor (CGM).
The G7 CGM is about the size of three stacked quarters and worn on the upper arm or abdomen. It sends Brooklyn glucose readings to a smartphone or watch every five minutes to keep track of blood sugar levels instead of making her prick her finger. She said the G7’s 30-minute warmup time and sensor grace period are her favorite features—no doubt to ensure a speedy return to the tennis court.
Brooklyn also recently updated the software on her t:slim X2 insulin pump to connect with her G7. This creates an automated insulin delivery (AID) system to predict and help prevent highs and lows. Of course, she must remember to input meals, periods of sleep, and tennis matches to keep up with her treatment.
Similarly, T’ara Smith has been living with diabetes since 2017 and wearing a t:slim X2 pump since 2022. The G7 and t:slim X2 integration also eased her management burden—she particularly enjoys setting an exercise activity timer for workouts.
“I’m able to set my settings for when I’m walking the local trails and when weightlifting,” she said.
The short warm-up period and 12-hour sensor grace period has eased her mind that she’s receiving the correct dosages of insulin. And Smith notes the t:slim X2’s “Control-IQ has been such a game-changer for me. Because of this device I feel like this diabetes has been more easy to manage.”
The ultimate goal for CGM and insulin pump technologies is to ease the diabetes struggle and manage the burdensome, chronic disease. Dexcom and Tandem are just two of the scientific and technological powerhouses working to ease the burden. Read on to learn more.
“Simplera Sync is an all-in-one, disposable, no fingerstick CGM that offers a simple, two-step insertion process with a discreet design without over tape,” said Ali Dianaty, SVP, product innovation and operation at Medtronic Diabetes. “The CGM is also integrated with our MiniMed 780G system, which automatically adjusts insulin every five minutes for users and delivers auto-corrections as needed, when an individual occasionally forgets to bolus or underestimates their carb count.”
The Simplera Sync CGM received EU approval to be linked with Medtronic’s MiniMed 780G system in January 2024, and will hit the European market via limited release this spring. The sensor leverages Medtronic’s advanced automatic insulin dosing (AID) algorithm in the MiniMed 780G system.
MiniMed 780G automatically adjusts and corrects glucose levels every five minutes. It has a Meal Detection feature designed to reduce post-meal hyperglycemia and is available as a seven-day infusion set. It features a glucose target setting as low as 100 mg/dL, which mirrors the glucose levels of someone without diabetes.
“The ADAPT study, which compared the MiniMed 780G system to standard of care (MDI + CGM), showed those using the MiniMed 780G system experienced a Time in Range increase of 27% over MDI (6.6 more hours/day in target range) and 1.4% reduction in A1c sustained out to one year in both the control and cross-over group,” said Dianaty. “We’re seeing growing bodies of clinical data that add to the proven benefits of our system. Despite the rapid adoption of CGM over the past decade, less than 30% of individuals on MDI therapy using a CGM achieve glycemic targets—highlighting a significant unmet need.”
In October 2023, Medtronic also presented multi-continent, real-world data that demonstrated using the MiniMed 780G system with its Guardian 4 CGM helps diabetes patients meet or exceed internationally recommended targets, regardless of the region of the world they reside in.
“The topic of hyperglycemia often gets overshadowed by discussion of lows, but there’s an urgent need to ensure awareness around the short- and long-term health complications which can impact just about every part of the body including organs, blood vessels, eyes, and even the brain,” said Dianaty. “The risk of these complications decreases with higher Time in Range and an A1c less than 7.7, so our goal is to enable more people to achieve tighter management without adding additional burden. With over 42 factors that impact blood sugar levels on any given day, it's maddening to think about how much you’d have to do to stay on top of this. Fewer than one in five people with type 1 diabetes are achieving blood sugar goals established by the American Diabetes Association, so we sought to find an easier way for people to stay on top of their highs while not exhausting themselves in the same way they’d have to if they tried to do this manually.”
“That’s why we’ve designed the MiniMed 780G system’s algorithm to take on more of the decision-making diabetes management requires on a day-to-day basis, and it’s performing incredibly well,” Dianaty went on. “Our goal with diabetes management is to get people as close to normoglycemia as possible, said another way—to get as close to the glycemic levels of someone not living with diabetes. Healthy individuals without diabetes spend about 96% of their day with blood glucose levels between 70-140 mg/dL—this is even tighter than the current metric of Time in Range (70-180 mg/dL) that’s used to measure good control in someone living with type 1 diabetes. It’s great that we’ve been able to come to a point in the industry where we’re raising the bar even higher, and why wouldn’t we?”
“Abbott’s acquisition of Bigfoot Biomedical brings together two important areas of diabetes management: continuous glucose monitoring and smart pen insulin dosing support,” an Abbott spokesperson told MPO. “By combining our companies, we can further develop connected solutions to help people living with diabetes by making diabetes management even more personal and precise.”
In January 2024, Abbott revealed that the t:slim X2 AID with Control-IQ was integrated with its FreeStyle Libre 2 Plus CGM. The FreeStyle Libre 2 Plus sensor is a modified version of the technology, and was cleared in 2023 by the FDA.
“FreeStyle Libre users in the U.S. now have the option to experience an automated insulin delivery (AID) system. The integration helps predict and prevent high and low blood sugar, while automatically adjusting insulin delivery and lessening the burden of care management. FreeStyle Libre 2 Plus is the first and only 15-day CGM sensor available in the U.S. today—and because users only need two sensors a month, it saves them time and money.”
The company also released a connected app with WeightWatchers to help diabetic patients understand how food and activity affect their glucose levels. Using the app, members of WeightWatchers following the WW Diabetes-Tailored Plan can access glucose data from the FreeStyle Libre 2 CGM system in their WW app while being guided by the plan. The company is also developing other types of sensor technologies.
“Abbott is currently developing a sensor that will measure both glucose and ketone levels at the same time. For people with diabetes, high ketone levels can be dangerous, or even fatal. High ketone levels are a result of dangerously low insulin levels, so the body begins accumulating ketones in the blood to compensate. The risk of this is most high in people who use meal-time insulin.”
“Today, if people with diabetes need to test their ketones, they can prick their finger or take a urine test,” they went on. “Most people don’t do this—mainly because they weren’t aware they needed to, but also because they don’t regularly keep these tests at home. The development of a dual glucose and ketone sensor will give people better peace of mind knowing what exactly is happening inside their body.”
“For diabetes patients who rely on insulin multiple times a day, Dexcom’s newest integration with the Tandem t:slim X2 insulin pump not only offers users more choice and flexibility when it comes to how they deliver insulin, but it also means they can now use the world’s most accurate CGM, Dexcom G7, to manage their diabetes,” Dexcom CEO Kevin Sayer told MPO. “The clinical benefits and increased peace of mind for users are clear. For instance, on a Tandem t:slim X2 insulin pump, Dexcom CGM users experience 11% more time in range. Furthermore, new results from the COMISAIR seven-year follow up—the longest prospective real-world CGM study ever conducted—also showed substantial reduction in HbA1c when a Dexcom CGM is connected to the Tandem t:slim X2 insulin pump with Control-IQ technology.”
In January 2024, the company announced a new glucose sensor that’s meant for type 2 diabetes patients who don’t use insulin. Dexcom expects the new sensor to launch in the U.S. this summer.
“Dexcom recently revealed that Stelo, its new glucose sensor, has been submitted to the FDA and will launch in the U.S. this summer,” Sayer said. “Stelo will offer 15-day sensor wear, a cash-pay option, and a software experience tailored specifically for people with Type 2 diabetes who don’t use insulin, addressing a huge unmet need for this population of around 25 million people in the U.S. Building on Dexcom’s 25-year history making CGM technology that has revolutionized diabetes management, Stelo will open up innovative tech and critical insights to a new population looking to prevent chronic disease progression.”
“As we continue to build our roadmap, one ultimate focus is providing people living with diabetes a choice when it comes to their diabetes management,” said Elizabeth Gasser, EVP and chief strategy officer at Tandem. “This commitment led to our efforts to expand the portfolio of continuous glucose monitoring (CGM) sensors that can be integrated with the t:slim X2.”
“With the Dexcom G7 integration, t:slim X2 users can now experience improved accuracy, a shorter warm-up time, and a 12-hour grace period that allows them to have a more seamless transition in between sessions,” Gasser went on. “That means they not only have the advantage of selecting a CGM that aligns with their preferences and lifestyle, but they also benefit from enhanced performance and convenience.”
February 2024 saw the release of Tandem Mobi, the company’s new AID system, in the U.S. According to the company, it’s the world’s smallest, durable AID system for diabetes patients—it’s less than half the size of the company’s t:slim X2 insulin pump.
“In order to meet the clinical needs of the diabetes population, we’re extending our business ventures beyond existing partnerships and rolling out innovative new solutions. We recognize that to provide our users with solutions that meet their personal needs, it’s important to provide them with options,” said Gasser. “That’s why we developed the Tandem Mobi, the world’s smallest automated insulin delivery (AID), to give users an even more discreet way to manage their diabetes. Less than half the size of the t:slim X2 and easily worn on-body, clipped to clothing, or stored in a coin pocket. Launched in February 2024, Tandem Mobi allows users increased flexibility and convenience.”
In December 2023, the company also launched Tandem Source, a diabetes management platform for insulin pump users and healthcare providers (HCPs). Tandem Source is designed to bring together the features of Tandem’s legacy t:connect, t:connect HCP, and t:connect Portal offerings with new comprehensive data reporting in one central, scalable platform.
“In addition to new solutions, we’re continuing to pursue partnerships with other innovators within the diabetes landscape. Earlier last year, we completed our acquisition of AMF Medical SA, the developer of the Sigi Patch Pump, an ergonomic and rechargeable tubeless pump for those managing type 1 diabetes. Our goal here is to broaden the range of users that we serve and provide more choice when it comes to their preferred method of insulin delivery.”
Tegra Medical, for instance, manufacturers components and assemblies for continuous glucose monitoring, drug delivery devices, and custom needles.
“The devices often require multiple technologies, such as micro molding, micro machining, and precision progressive stamping—all of which are within our capabilities,” said Mike Treleaven, senior VP of engineering and Darren Vine, director of European sales and global marketing, at Tegra Medical. “Our manufacturing locations in Switzerland and Asia specialize in these capabilities, therefore we can engage with companies searching for our level of expertise. Having these centers of excellence across the globe makes us the perfect partner for these dynamic growth opportunities in diabetes.”
When a diabetes device maker needs specialized support for any number of the components or manufacturing services that their devices require, they often turn to partners like Tegra to fill the gaps.
“Manufacturing technologies require expensive capital equipment and highly trained support staff, which are difficult for OEMs to maintain. Contract manufacturers like us already have the equipment, skills, know how, and trained labor to create their medical devices.”
Treleaven and Vine also commented on trends in the market they are observing.
“The trend we see is the increase in customers looking for components for continuous glucose monitoring, auto injection, and wearables,” they said.
Brusco: What are the trends right now in continuous glucose monitoring (CGM) technology?
Mona Eskandarian: Different CGM technologies and vendors available commercially are also utilized in clinical research. The devices vary very much when it comes to capabilities: sensor life (10 days vs. 14 days); how data is obtained from the CGM (through a reader or in real-time via an app); and the device’s recording intervals over the sensor life (every five or 15 minutes). When choosing which device to use in a clinical trial, assess the best suitable device based on the protocol requirements and the commercial device’s geographic availability.
Earlier generations of CGM devices required patients to scan the sensor with a reader at certain intervals to collect data and upload to a computer for readout; otherwise, data would be lost. Now, we see more use of CGM devices where sensor data is transmitted without human interaction (automatically through Bluetooth to an app on the patient’s mobile phone). By partnering with vendors providing digital health platforms, the app can be configured to allow or disable the patient’s view of their data, so participants in a clinical study will not have access to data if they are blinded.
In clinical research, the use of CGM devices is especially effective in decentralized clinical trials, where patient data can be collected remotely and uploaded with minimal human interaction. CGM technology is mainly used in Type 1 and 2 diabetes clinical studies where secondary endpoints and efficacy are assessed by examining time-in-range values before and after treatment. Real-time CGM data are also used to support dose titration/adjustments while participating in a clinical study. There is also a move toward using CGM in non-diabetes studies where certain glucose targets are set to enforce lifestyle changes; for example, in obesity studies or with high-performance athletes.
CGMs generate a lot of data—over 1,300 data points per visit—when used in clinical research. Proper analysis and review of the data generated from CGMs requires use of data “lakes” to create dashboards that contain visualizations to use for safety monitoring and identification of hypoglycemic events, compliance monitoring, and scientific surveillance on the research data.
When reviewing CGM data, it is important to understand how the data collected from a CGM wearable can be impacted and to ensure correct interpretation when compared to glucose values from a traditional finger prick in diabetes studies. CGM and finger prick or plasma glucose measurements may not always agree. This is due to the gradient in glucose levels that may exist between the two different compartments where the glucose measurements are made (interstitial space for CGM vs. plasma or blood).
In addition, external pressure from a lounge chair or bed to the site where the CGM sensor is inserted can cause low values due to decreased blood flow to a pressured area. This should not be considered a fault, since the sensor actually measures the true glucose value in interstitial fluid in subcutaneous tissue at that point in time. CGM data can also be impacted by patient use, for example—sensor displaced, sensor deliberately removed or sensor failure, resulting in lower device compliance.
Lastly, when using a device where CGM data is transmitted automatically to an app on a mobile phone, connectivity is critical to ensuring continuous data can be collected. Proactive measures to verify and mitigate these challenges are important for the success of the trial.”
Brusco: How does integration of CGM with insulin pump technology advance diabetes management strategy?
Eskandarian: Recently, various hybrid closed-loop systems have become commercially available. These systems maintain glucose levels within a target range through use of a computerized algorithm to adjust the basal rate of insulin and administer corrective bolus doses. They are called “hybrid” systems because, unlike, fully closed-loop systems, the user is still required to manually program insulin boluses with meals.”
Another development is dual hormone systems, where there’s an incorporation of glucagon and insulin. Use of these closed-loop systems is not without risk, as they have not undergone extensive testing, nor are they approved by the FDA.
Thankfully, modern medicine has the technology to help young Brooklyn manage her illness so she can spend more time doing the things she loves—and less time worrying about glucose numbers and insulin injections. She wears both the Tandem t:slim X2 insulin pump and Dexcom G7 continuous glucose monitor (CGM).
The G7 CGM is about the size of three stacked quarters and worn on the upper arm or abdomen. It sends Brooklyn glucose readings to a smartphone or watch every five minutes to keep track of blood sugar levels instead of making her prick her finger. She said the G7’s 30-minute warmup time and sensor grace period are her favorite features—no doubt to ensure a speedy return to the tennis court.
Brooklyn also recently updated the software on her t:slim X2 insulin pump to connect with her G7. This creates an automated insulin delivery (AID) system to predict and help prevent highs and lows. Of course, she must remember to input meals, periods of sleep, and tennis matches to keep up with her treatment.
Similarly, T’ara Smith has been living with diabetes since 2017 and wearing a t:slim X2 pump since 2022. The G7 and t:slim X2 integration also eased her management burden—she particularly enjoys setting an exercise activity timer for workouts.
“I’m able to set my settings for when I’m walking the local trails and when weightlifting,” she said.
The short warm-up period and 12-hour sensor grace period has eased her mind that she’s receiving the correct dosages of insulin. And Smith notes the t:slim X2’s “Control-IQ has been such a game-changer for me. Because of this device I feel like this diabetes has been more easy to manage.”
The ultimate goal for CGM and insulin pump technologies is to ease the diabetes struggle and manage the burdensome, chronic disease. Dexcom and Tandem are just two of the scientific and technological powerhouses working to ease the burden. Read on to learn more.
Medtronic
In September 2023, Medtronic earned CE mark approval for its new Simplera Sync continuous glucose monitor (CGM). The disposable CGM device linked with InPen, the company’s smart insulin pen. InPen consists of a reusable smart pen and a Bluetooth-enabled pen to calculate insulin doses, provide dose reminders, track active insulin, and send reports to caregivers.“Simplera Sync is an all-in-one, disposable, no fingerstick CGM that offers a simple, two-step insertion process with a discreet design without over tape,” said Ali Dianaty, SVP, product innovation and operation at Medtronic Diabetes. “The CGM is also integrated with our MiniMed 780G system, which automatically adjusts insulin every five minutes for users and delivers auto-corrections as needed, when an individual occasionally forgets to bolus or underestimates their carb count.”
The Simplera Sync CGM received EU approval to be linked with Medtronic’s MiniMed 780G system in January 2024, and will hit the European market via limited release this spring. The sensor leverages Medtronic’s advanced automatic insulin dosing (AID) algorithm in the MiniMed 780G system.
MiniMed 780G automatically adjusts and corrects glucose levels every five minutes. It has a Meal Detection feature designed to reduce post-meal hyperglycemia and is available as a seven-day infusion set. It features a glucose target setting as low as 100 mg/dL, which mirrors the glucose levels of someone without diabetes.
“The ADAPT study, which compared the MiniMed 780G system to standard of care (MDI + CGM), showed those using the MiniMed 780G system experienced a Time in Range increase of 27% over MDI (6.6 more hours/day in target range) and 1.4% reduction in A1c sustained out to one year in both the control and cross-over group,” said Dianaty. “We’re seeing growing bodies of clinical data that add to the proven benefits of our system. Despite the rapid adoption of CGM over the past decade, less than 30% of individuals on MDI therapy using a CGM achieve glycemic targets—highlighting a significant unmet need.”
In October 2023, Medtronic also presented multi-continent, real-world data that demonstrated using the MiniMed 780G system with its Guardian 4 CGM helps diabetes patients meet or exceed internationally recommended targets, regardless of the region of the world they reside in.
“The topic of hyperglycemia often gets overshadowed by discussion of lows, but there’s an urgent need to ensure awareness around the short- and long-term health complications which can impact just about every part of the body including organs, blood vessels, eyes, and even the brain,” said Dianaty. “The risk of these complications decreases with higher Time in Range and an A1c less than 7.7, so our goal is to enable more people to achieve tighter management without adding additional burden. With over 42 factors that impact blood sugar levels on any given day, it's maddening to think about how much you’d have to do to stay on top of this. Fewer than one in five people with type 1 diabetes are achieving blood sugar goals established by the American Diabetes Association, so we sought to find an easier way for people to stay on top of their highs while not exhausting themselves in the same way they’d have to if they tried to do this manually.”
“That’s why we’ve designed the MiniMed 780G system’s algorithm to take on more of the decision-making diabetes management requires on a day-to-day basis, and it’s performing incredibly well,” Dianaty went on. “Our goal with diabetes management is to get people as close to normoglycemia as possible, said another way—to get as close to the glycemic levels of someone not living with diabetes. Healthy individuals without diabetes spend about 96% of their day with blood glucose levels between 70-140 mg/dL—this is even tighter than the current metric of Time in Range (70-180 mg/dL) that’s used to measure good control in someone living with type 1 diabetes. It’s great that we’ve been able to come to a point in the industry where we’re raising the bar even higher, and why wouldn’t we?”
Abbott
In September 2023, Abbott Labs completed its acquisition of Bigfoot Biomedical, a developer of smart insulin management systems for patient with diabetes. Bigfoot’s Unity system exclusively works with Abbott’s FreeStyle Libre tech and includes a customer smartphone app connected to a cloud-based online portal used by healthcare providers to support their patients, including remote care.“Abbott’s acquisition of Bigfoot Biomedical brings together two important areas of diabetes management: continuous glucose monitoring and smart pen insulin dosing support,” an Abbott spokesperson told MPO. “By combining our companies, we can further develop connected solutions to help people living with diabetes by making diabetes management even more personal and precise.”
In January 2024, Abbott revealed that the t:slim X2 AID with Control-IQ was integrated with its FreeStyle Libre 2 Plus CGM. The FreeStyle Libre 2 Plus sensor is a modified version of the technology, and was cleared in 2023 by the FDA.
“FreeStyle Libre users in the U.S. now have the option to experience an automated insulin delivery (AID) system. The integration helps predict and prevent high and low blood sugar, while automatically adjusting insulin delivery and lessening the burden of care management. FreeStyle Libre 2 Plus is the first and only 15-day CGM sensor available in the U.S. today—and because users only need two sensors a month, it saves them time and money.”
The company also released a connected app with WeightWatchers to help diabetic patients understand how food and activity affect their glucose levels. Using the app, members of WeightWatchers following the WW Diabetes-Tailored Plan can access glucose data from the FreeStyle Libre 2 CGM system in their WW app while being guided by the plan. The company is also developing other types of sensor technologies.
“Abbott is currently developing a sensor that will measure both glucose and ketone levels at the same time. For people with diabetes, high ketone levels can be dangerous, or even fatal. High ketone levels are a result of dangerously low insulin levels, so the body begins accumulating ketones in the blood to compensate. The risk of this is most high in people who use meal-time insulin.”
“Today, if people with diabetes need to test their ketones, they can prick their finger or take a urine test,” they went on. “Most people don’t do this—mainly because they weren’t aware they needed to, but also because they don’t regularly keep these tests at home. The development of a dual glucose and ketone sensor will give people better peace of mind knowing what exactly is happening inside their body.”
Dexcom
In December 2023, the Dexcom G7 CGM was launched as an integrated system with Tandem Diabetes Care’s t:slim X2 automated insulin delivery (AID) system in the U.S. Tandem’s AID system with Control-IQ technology was the first AID option to use Dexcom’s latest CGM technology.“For diabetes patients who rely on insulin multiple times a day, Dexcom’s newest integration with the Tandem t:slim X2 insulin pump not only offers users more choice and flexibility when it comes to how they deliver insulin, but it also means they can now use the world’s most accurate CGM, Dexcom G7, to manage their diabetes,” Dexcom CEO Kevin Sayer told MPO. “The clinical benefits and increased peace of mind for users are clear. For instance, on a Tandem t:slim X2 insulin pump, Dexcom CGM users experience 11% more time in range. Furthermore, new results from the COMISAIR seven-year follow up—the longest prospective real-world CGM study ever conducted—also showed substantial reduction in HbA1c when a Dexcom CGM is connected to the Tandem t:slim X2 insulin pump with Control-IQ technology.”
In January 2024, the company announced a new glucose sensor that’s meant for type 2 diabetes patients who don’t use insulin. Dexcom expects the new sensor to launch in the U.S. this summer.
“Dexcom recently revealed that Stelo, its new glucose sensor, has been submitted to the FDA and will launch in the U.S. this summer,” Sayer said. “Stelo will offer 15-day sensor wear, a cash-pay option, and a software experience tailored specifically for people with Type 2 diabetes who don’t use insulin, addressing a huge unmet need for this population of around 25 million people in the U.S. Building on Dexcom’s 25-year history making CGM technology that has revolutionized diabetes management, Stelo will open up innovative tech and critical insights to a new population looking to prevent chronic disease progression.”
Tandem Diabetes Care
As previously mentioned, Tandem Diabetes released updated t:slim X2 insulin pump software with Dexcom G7 CGM integration in the U.S. in December 2023. Providing a perspective from the other side of the partnership, Tandem Diabetes executives spoke about the benefits of the integration for diabetes patients.“As we continue to build our roadmap, one ultimate focus is providing people living with diabetes a choice when it comes to their diabetes management,” said Elizabeth Gasser, EVP and chief strategy officer at Tandem. “This commitment led to our efforts to expand the portfolio of continuous glucose monitoring (CGM) sensors that can be integrated with the t:slim X2.”
“With the Dexcom G7 integration, t:slim X2 users can now experience improved accuracy, a shorter warm-up time, and a 12-hour grace period that allows them to have a more seamless transition in between sessions,” Gasser went on. “That means they not only have the advantage of selecting a CGM that aligns with their preferences and lifestyle, but they also benefit from enhanced performance and convenience.”
February 2024 saw the release of Tandem Mobi, the company’s new AID system, in the U.S. According to the company, it’s the world’s smallest, durable AID system for diabetes patients—it’s less than half the size of the company’s t:slim X2 insulin pump.
“In order to meet the clinical needs of the diabetes population, we’re extending our business ventures beyond existing partnerships and rolling out innovative new solutions. We recognize that to provide our users with solutions that meet their personal needs, it’s important to provide them with options,” said Gasser. “That’s why we developed the Tandem Mobi, the world’s smallest automated insulin delivery (AID), to give users an even more discreet way to manage their diabetes. Less than half the size of the t:slim X2 and easily worn on-body, clipped to clothing, or stored in a coin pocket. Launched in February 2024, Tandem Mobi allows users increased flexibility and convenience.”
In December 2023, the company also launched Tandem Source, a diabetes management platform for insulin pump users and healthcare providers (HCPs). Tandem Source is designed to bring together the features of Tandem’s legacy t:connect, t:connect HCP, and t:connect Portal offerings with new comprehensive data reporting in one central, scalable platform.
“In addition to new solutions, we’re continuing to pursue partnerships with other innovators within the diabetes landscape. Earlier last year, we completed our acquisition of AMF Medical SA, the developer of the Sigi Patch Pump, an ergonomic and rechargeable tubeless pump for those managing type 1 diabetes. Our goal here is to broaden the range of users that we serve and provide more choice when it comes to their preferred method of insulin delivery.”
Manufacturing Support
Like a good portion of the medical device industry, smaller and smarter is a major trend affecting diabetes technology. Some of the components and manufacturing methods needed are simply not within the device maker’s bandwidth. When this occurs, these organizations may turn to a manufacturing partner with specialized expertise to help manufacture certain parts of their device.Tegra Medical, for instance, manufacturers components and assemblies for continuous glucose monitoring, drug delivery devices, and custom needles.
“The devices often require multiple technologies, such as micro molding, micro machining, and precision progressive stamping—all of which are within our capabilities,” said Mike Treleaven, senior VP of engineering and Darren Vine, director of European sales and global marketing, at Tegra Medical. “Our manufacturing locations in Switzerland and Asia specialize in these capabilities, therefore we can engage with companies searching for our level of expertise. Having these centers of excellence across the globe makes us the perfect partner for these dynamic growth opportunities in diabetes.”
When a diabetes device maker needs specialized support for any number of the components or manufacturing services that their devices require, they often turn to partners like Tegra to fill the gaps.
“Manufacturing technologies require expensive capital equipment and highly trained support staff, which are difficult for OEMs to maintain. Contract manufacturers like us already have the equipment, skills, know how, and trained labor to create their medical devices.”
Treleaven and Vine also commented on trends in the market they are observing.
“The trend we see is the increase in customers looking for components for continuous glucose monitoring, auto injection, and wearables,” they said.
Continuous Glucose Monitoringin Clinical Research
To gain more insights about broader uses for CGM and smart insulin pump technology, MPO spoke to Mona Eskandarian, director of product management, general medicine and cardiovascular, at PPD. PPD, part of Thermo Fisher Scientific, is a strategic partner in clinical development and analytical services.Brusco: What are the trends right now in continuous glucose monitoring (CGM) technology?
Mona Eskandarian: Different CGM technologies and vendors available commercially are also utilized in clinical research. The devices vary very much when it comes to capabilities: sensor life (10 days vs. 14 days); how data is obtained from the CGM (through a reader or in real-time via an app); and the device’s recording intervals over the sensor life (every five or 15 minutes). When choosing which device to use in a clinical trial, assess the best suitable device based on the protocol requirements and the commercial device’s geographic availability.
Earlier generations of CGM devices required patients to scan the sensor with a reader at certain intervals to collect data and upload to a computer for readout; otherwise, data would be lost. Now, we see more use of CGM devices where sensor data is transmitted without human interaction (automatically through Bluetooth to an app on the patient’s mobile phone). By partnering with vendors providing digital health platforms, the app can be configured to allow or disable the patient’s view of their data, so participants in a clinical study will not have access to data if they are blinded.
In clinical research, the use of CGM devices is especially effective in decentralized clinical trials, where patient data can be collected remotely and uploaded with minimal human interaction. CGM technology is mainly used in Type 1 and 2 diabetes clinical studies where secondary endpoints and efficacy are assessed by examining time-in-range values before and after treatment. Real-time CGM data are also used to support dose titration/adjustments while participating in a clinical study. There is also a move toward using CGM in non-diabetes studies where certain glucose targets are set to enforce lifestyle changes; for example, in obesity studies or with high-performance athletes.
CGMs generate a lot of data—over 1,300 data points per visit—when used in clinical research. Proper analysis and review of the data generated from CGMs requires use of data “lakes” to create dashboards that contain visualizations to use for safety monitoring and identification of hypoglycemic events, compliance monitoring, and scientific surveillance on the research data.
When reviewing CGM data, it is important to understand how the data collected from a CGM wearable can be impacted and to ensure correct interpretation when compared to glucose values from a traditional finger prick in diabetes studies. CGM and finger prick or plasma glucose measurements may not always agree. This is due to the gradient in glucose levels that may exist between the two different compartments where the glucose measurements are made (interstitial space for CGM vs. plasma or blood).
In addition, external pressure from a lounge chair or bed to the site where the CGM sensor is inserted can cause low values due to decreased blood flow to a pressured area. This should not be considered a fault, since the sensor actually measures the true glucose value in interstitial fluid in subcutaneous tissue at that point in time. CGM data can also be impacted by patient use, for example—sensor displaced, sensor deliberately removed or sensor failure, resulting in lower device compliance.
Lastly, when using a device where CGM data is transmitted automatically to an app on a mobile phone, connectivity is critical to ensuring continuous data can be collected. Proactive measures to verify and mitigate these challenges are important for the success of the trial.”
Brusco: How does integration of CGM with insulin pump technology advance diabetes management strategy?
Eskandarian: Recently, various hybrid closed-loop systems have become commercially available. These systems maintain glucose levels within a target range through use of a computerized algorithm to adjust the basal rate of insulin and administer corrective bolus doses. They are called “hybrid” systems because, unlike, fully closed-loop systems, the user is still required to manually program insulin boluses with meals.”
Another development is dual hormone systems, where there’s an incorporation of glucagon and insulin. Use of these closed-loop systems is not without risk, as they have not undergone extensive testing, nor are they approved by the FDA.