Michael Barbella, Managing Editor01.16.24
RF Medical Co. Ltd. has successfully completed the transition to the European Medical Device Regulation (MDR) CE Certification.
The European Medical Device Regulation, also known as CE MDR, strengthens the existing European Medical Device Directive (CE MDD). The requirements for product quality, safety, performance effectiveness, and clinical evaluation have become more stringent under the MDR. Medical device manufacturers wishing to commercialize their products in Europe must comply with this regulation.
An official from RF Medical commented, "We are delighted to have reached another significant milestone in RF Medical's 20-year journey of developing and commercializing minimally invasive ablation devices. Thanks to our commitment to quality enhancement and system improvement, the company can continue to align with global standards."
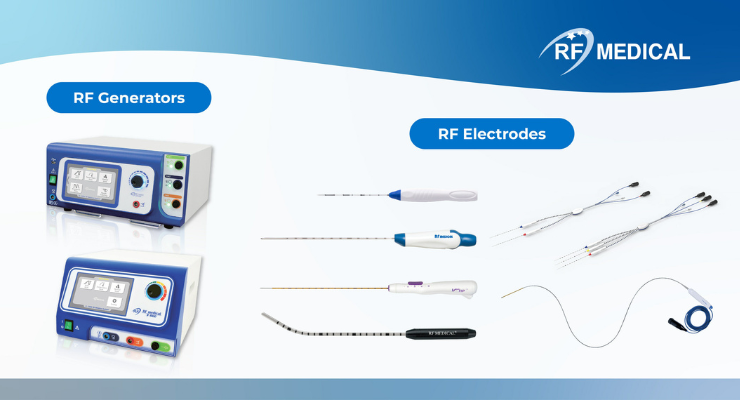
The MDR certificate encompasses all of RF Medical's product portfolios previously compliant with the MDD. This includes RF generators, such as MYGEN M-3004, MYGEN V-1000, and VeinCLEAR V-700, as well as RF electrodes including Big-Tip, JET-TIP, THYBLATE, MYOBLATE, and VeinCLEAR catheters. The acquisition of this certificate ensures the continuous supply of these products to the European market.
RF Medical's transition to the new regulations not only positions the company for sustained success in the global medical device market but also underscores its dedication to delivering reliable medical solutions to healthcare practitioners and patients.
South Korea-based RF Medical manufactures minimally invasive solutions for tumor treatment. With advanced technology and proven quality, the company has extended its product reach to more than 60 countries worldwide. As a medical device company, RF Medical is dedicated to continuous investments in a superior workforce, manufacturing processes, innovation, and research and development. Pioneering the future, the firm is committed to developing effective, affordable, and less-invasive medical technologies.
The European Medical Device Regulation, also known as CE MDR, strengthens the existing European Medical Device Directive (CE MDD). The requirements for product quality, safety, performance effectiveness, and clinical evaluation have become more stringent under the MDR. Medical device manufacturers wishing to commercialize their products in Europe must comply with this regulation.
An official from RF Medical commented, "We are delighted to have reached another significant milestone in RF Medical's 20-year journey of developing and commercializing minimally invasive ablation devices. Thanks to our commitment to quality enhancement and system improvement, the company can continue to align with global standards."
The MDR certificate encompasses all of RF Medical's product portfolios previously compliant with the MDD. This includes RF generators, such as MYGEN M-3004, MYGEN V-1000, and VeinCLEAR V-700, as well as RF electrodes including Big-Tip, JET-TIP, THYBLATE, MYOBLATE, and VeinCLEAR catheters. The acquisition of this certificate ensures the continuous supply of these products to the European market.
RF Medical's transition to the new regulations not only positions the company for sustained success in the global medical device market but also underscores its dedication to delivering reliable medical solutions to healthcare practitioners and patients.
South Korea-based RF Medical manufactures minimally invasive solutions for tumor treatment. With advanced technology and proven quality, the company has extended its product reach to more than 60 countries worldwide. As a medical device company, RF Medical is dedicated to continuous investments in a superior workforce, manufacturing processes, innovation, and research and development. Pioneering the future, the firm is committed to developing effective, affordable, and less-invasive medical technologies.