Sam Brusco, Associate Editor01.11.24
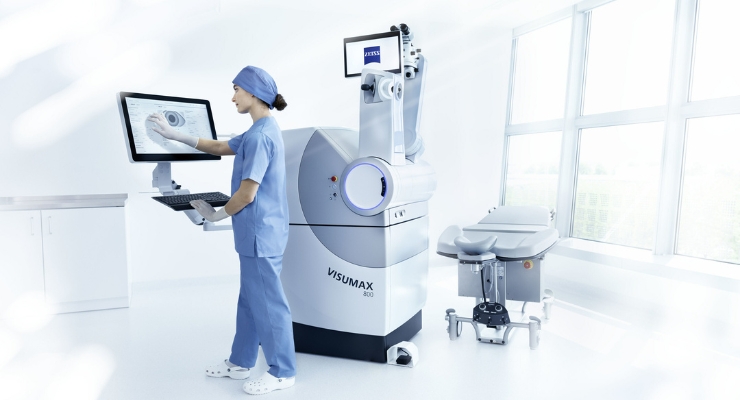
ZEISS Medical Technology has earned U.S. Food and Drug Administration (FDA) approval for the VisuMax 800 with ZEISS Smile pro software to surgically treat nearsightedness, with or without astigmatism.
VisuMax 800 with Smile pro creates the lenticule in under 10 seconds because of a faster femtosecond laser pulse repetition of 2 MHz. The laser also has a smaller footprint and compatibility with a variety of patient beds, adapting to the clinical environment as needed.
The system includes the CentraLign centration aid, a computer-controlled function for easier centration. Its OcuLign feature helps counter potential cyclotorsion, and Visulyze user nomograms help to collect and analyze patient data as well as provide detailed nomograms.
"ZEISS continues to set itself apart in the U.S. market with the availability of the VISUMAX 800 with SMILE pro software from ZEISS, providing the latest digital technology from the company's legacy of innovation to meet the evolving needs of refractive surgeons," said Euan S. Thomson, Ph.D., president of Ophthalmology Strategic Business Unit and Head of the Digital Business Unit for ZEISS Medical Technology. "As part of the ZEISS Medical Ecosystem, this next generation femtosecond laser system creates data-driven insights to help surgeons manage better treatment paths for patients while supporting each surgeon's unique practice requirements for greater workflow efficiency and performance."
With these workflow enhancements along with greater flexibility and faster treatment, the VISUMAX 800 with SMILE pro software from ZEISS offers substantial market opportunities for U.S. surgeons. "The VISUMAX 800 is a practice builder and allows surgeons to differentiate their practice from local competitors," added Luke Rebenitsch, MD, at ClearSight LASIK and 43Vision, in Oklahoma City.
Last month, Carl Zeiss Meditec began a deal to acquire 100% of the shares in Dutch Ophthalmic Research Center (D.O.R.C.).
VisuMax 800 with Smile pro creates the lenticule in under 10 seconds because of a faster femtosecond laser pulse repetition of 2 MHz. The laser also has a smaller footprint and compatibility with a variety of patient beds, adapting to the clinical environment as needed.
The system includes the CentraLign centration aid, a computer-controlled function for easier centration. Its OcuLign feature helps counter potential cyclotorsion, and Visulyze user nomograms help to collect and analyze patient data as well as provide detailed nomograms.
"ZEISS continues to set itself apart in the U.S. market with the availability of the VISUMAX 800 with SMILE pro software from ZEISS, providing the latest digital technology from the company's legacy of innovation to meet the evolving needs of refractive surgeons," said Euan S. Thomson, Ph.D., president of Ophthalmology Strategic Business Unit and Head of the Digital Business Unit for ZEISS Medical Technology. "As part of the ZEISS Medical Ecosystem, this next generation femtosecond laser system creates data-driven insights to help surgeons manage better treatment paths for patients while supporting each surgeon's unique practice requirements for greater workflow efficiency and performance."
With these workflow enhancements along with greater flexibility and faster treatment, the VISUMAX 800 with SMILE pro software from ZEISS offers substantial market opportunities for U.S. surgeons. "The VISUMAX 800 is a practice builder and allows surgeons to differentiate their practice from local competitors," added Luke Rebenitsch, MD, at ClearSight LASIK and 43Vision, in Oklahoma City.
Last month, Carl Zeiss Meditec began a deal to acquire 100% of the shares in Dutch Ophthalmic Research Center (D.O.R.C.).