Sam Brusco, Associate Editor12.15.23
Fresenius Kabi has signed a multiple year agreement with the Mayo Clinic, under which the Mayo Clinic is expected to buy 10,000 of the company’s Ivenix large-volume infusion pumps for its hospitals and clinics in Minnesota, Arizona, and Florida.
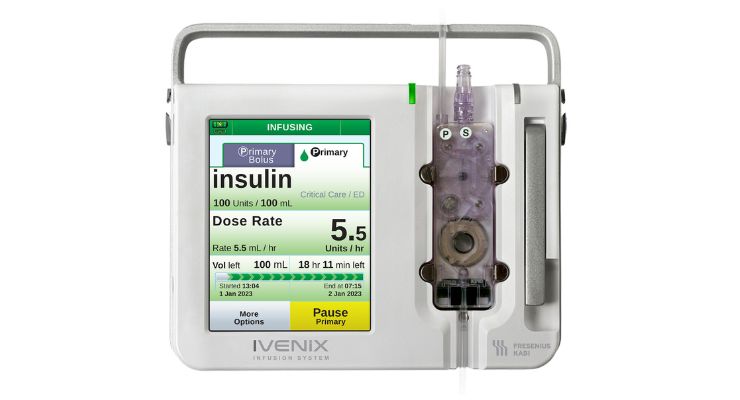
The Ivenix infusion system is a “smart pump” with a smartphone-like touchscreen and patented pumping mechanism. The pumps also can interface with hospital information systems and electronic medical records.
Fresenius Kabi said this is the biggest contract for Ivenix pumps the company has signed to date.
“Technology is transforming patient care, and at Fresenius Kabi our focus is on scaling advanced technologies and essential medicines so they can benefit more patients in more places,” said Dr. Christian Hauer, president of Fresenius Kabi MedTech. “We are honored to collaborate with—and to serve—the care teams at Mayo.”
The company bought Ivenix last March for $220 million. The large volume pump features administration sets, infusion management software tools, applications, and analytics. The system was designed to minimize infusion-related errors and lower total cost of ownership.
The infusion system launched in late 2021, after receiving U.S. Food and Drug Administration (FDA) approval.
The Ivenix infusion system is a “smart pump” with a smartphone-like touchscreen and patented pumping mechanism. The pumps also can interface with hospital information systems and electronic medical records.
Fresenius Kabi said this is the biggest contract for Ivenix pumps the company has signed to date.
“Technology is transforming patient care, and at Fresenius Kabi our focus is on scaling advanced technologies and essential medicines so they can benefit more patients in more places,” said Dr. Christian Hauer, president of Fresenius Kabi MedTech. “We are honored to collaborate with—and to serve—the care teams at Mayo.”
The company bought Ivenix last March for $220 million. The large volume pump features administration sets, infusion management software tools, applications, and analytics. The system was designed to minimize infusion-related errors and lower total cost of ownership.
The infusion system launched in late 2021, after receiving U.S. Food and Drug Administration (FDA) approval.