Sam Brusco, Associate Editor07.24.23
BD (Becton Dickinson) has gained U.S. Food and Drug Administration (FDA) 510(k) clearance for its updated Alaris infusion system, enabling remediation and return to full commercial operations for the infusion system in the U.S.
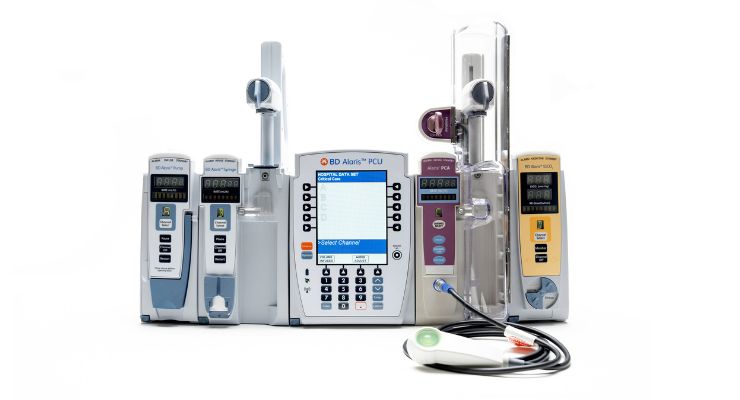
The FDA nod covers updated hardware features for Point-of-Care Unit (PCU), large volume pumps, syringe pumps, respiratory monitoring, and auto-identification modules. It also covers a new software version with better cybersecurity, according to the company.
The new Alaris software also has interoperability features to allow smart, connected care with the most common electronic medical record (EMR) systems.
The Alaris infusion pump has been subject to a number of recalls over the past few years. In 2020, BD began a Class I recall that was caused by multiple system, software, and use-related errors. The company then started a hold of new Alaris pump shipments. BD applied for a new FDA clearance for Alaris in April 2021, and began remediation efforts two months later.
Earlier this year, BD released a notification concerning cybersecurity weaknesses in its Alaris Infusion Central software, and last week another notification was issued that warned about eight cybersecurity vulnerabilities.
"The 510(k) clearance of the updated BD Alaris Infusion System has been the number one priority for our teams who have been steadfast in their efforts to achieve this milestone, consistent with our commitment to quality," Tom Polen, chairman, CEO, and president of BD told the press. "We are deeply committed to ensuring clinicians can continue to rely on our market leading system to meet today's most critical infusion needs. The features and enhancements incorporated into the updated BD Alaris Infusion System and subject to this clearance reinforce our advancements in smart, connected care, which deliver greater benefit to clinicians and patients and help improve health care system efficiency through better care coordination and utilization of actionable information."
BD touts Alaris as the only system with a centralized user interface for up to four modules. This covers all major types of infusions: large volume pumps, syringe pumps, and PCA therapy with optional respiratory monitoring.
The company also says it has the largest breadth of customers, nurses trained on its use, and number of customers with EMR interoperability. Its digital connectivity can help to avoid navigating time-consuming processes and multiple steps to manually program infusions.
The company is addressing all open recalls by remediating or replacing all current U.S. Alaris devices with the updated software. BD said the large installed Alaris base, it was start working in close partnership with customers.
BD said it’s been preparing for the updated clearance and returning to full commercial operations by investing in operational capacity and functional capabilities. This included investments to boost manufacturing capacity, strengthen supply chains, and increase supplier redundancy.
"Remediation of recalls will always be a top priority for BD, and our remediation plans go above and beyond all open recall requirements to ensure all devices are compliant with the 510(k) clearance, including the newest hardware, software and cybersecurity updates," said Ami Simunovich, chief quality and regulatory officer at BD. "We have made substantial investments to simplify, improve and evolve our processes, systems and policies to ensure we design, manufacture and sustain our products in compliance with changing global regulatory and safety requirements. We remain committed to delivering next-generation quality and compliance to our customers."
The FDA nod covers updated hardware features for Point-of-Care Unit (PCU), large volume pumps, syringe pumps, respiratory monitoring, and auto-identification modules. It also covers a new software version with better cybersecurity, according to the company.
The new Alaris software also has interoperability features to allow smart, connected care with the most common electronic medical record (EMR) systems.
The Alaris infusion pump has been subject to a number of recalls over the past few years. In 2020, BD began a Class I recall that was caused by multiple system, software, and use-related errors. The company then started a hold of new Alaris pump shipments. BD applied for a new FDA clearance for Alaris in April 2021, and began remediation efforts two months later.
Earlier this year, BD released a notification concerning cybersecurity weaknesses in its Alaris Infusion Central software, and last week another notification was issued that warned about eight cybersecurity vulnerabilities.
"The 510(k) clearance of the updated BD Alaris Infusion System has been the number one priority for our teams who have been steadfast in their efforts to achieve this milestone, consistent with our commitment to quality," Tom Polen, chairman, CEO, and president of BD told the press. "We are deeply committed to ensuring clinicians can continue to rely on our market leading system to meet today's most critical infusion needs. The features and enhancements incorporated into the updated BD Alaris Infusion System and subject to this clearance reinforce our advancements in smart, connected care, which deliver greater benefit to clinicians and patients and help improve health care system efficiency through better care coordination and utilization of actionable information."
BD touts Alaris as the only system with a centralized user interface for up to four modules. This covers all major types of infusions: large volume pumps, syringe pumps, and PCA therapy with optional respiratory monitoring.
The company also says it has the largest breadth of customers, nurses trained on its use, and number of customers with EMR interoperability. Its digital connectivity can help to avoid navigating time-consuming processes and multiple steps to manually program infusions.
The company is addressing all open recalls by remediating or replacing all current U.S. Alaris devices with the updated software. BD said the large installed Alaris base, it was start working in close partnership with customers.
BD said it’s been preparing for the updated clearance and returning to full commercial operations by investing in operational capacity and functional capabilities. This included investments to boost manufacturing capacity, strengthen supply chains, and increase supplier redundancy.
"Remediation of recalls will always be a top priority for BD, and our remediation plans go above and beyond all open recall requirements to ensure all devices are compliant with the 510(k) clearance, including the newest hardware, software and cybersecurity updates," said Ami Simunovich, chief quality and regulatory officer at BD. "We have made substantial investments to simplify, improve and evolve our processes, systems and policies to ensure we design, manufacture and sustain our products in compliance with changing global regulatory and safety requirements. We remain committed to delivering next-generation quality and compliance to our customers."