Michael Barbella, Managing Editor07.21.23
CorFlow Therapeutics AG has announced interim positive study results for its CoFI System.
The company's MOCA I (Microvascular Obstruction with CoFI System Assessment) study is evaluating the safety and feasibility of the real-time catheter-based system to detect coronary microvascular obstruction MVO immediately after primary percutaneous coronary intervention (PPCI) and deliver timely therapeutic intracoronary agents. The trial data set included 30 patients with an ST elevation myocardial infarction (STEMI), the most severe type of heart attack.
“The data presented from the MOCA I study with the CorFlow system is very encouraging for our community of cardiologists, who are increasingly recognizing MVO and microvascular disease as a next frontier to improve outcomes in our field,” stated Dr. Azeem Latib of Montefiore Medical Center, New York, N.Y. “This emerging area is in its early days, but we can agree that a significant step forward is made by being able to diagnose these patients in the cath lab. With such a technology, we can imagine that many concomitant therapy options can soon be evaluated, since it is now clear that treating only the larger vessels is not enough.”
The system outputs a novel pressure parameter (PCoFI) for detection of MVO, which was evaluated for correlation with cardiac MRI measured MVO, considered the reference standard. For the patients who had the CoFI diagnostic sequence, the essential results of the study showed that the system provided a 95% sensitivity, 91% specificity, and 0.94 AUC for the ability to detect MVO measured by cardiac MRI. The CoFI device and procedure were deemed safe with no device-related deaths, flow-limiting dissections, or thrombotic events at 30 days, as adjudicated by an independent Clinical Events Committee.
The MOCA I study suggests that it might now be possible to have accurate, early detection which can lead to targeted treatment for these high-risk patients.
Current interventional cardiology technologies, addressing the larger coronary arteries, have been instrumental in saving patients from death and complications after a heart attack, reducing short term mortality over the last several decades. Yet for many patients, longer term outcomes are still poor despite current standard of care treatment. MVO, which is found to be in more than 50% of heart attack patients, has been shown to be the single most powerful prognostic indicator for negative outcomes for these patients, including heart failure and death. Heart disease remains the leading cause of death worldwide, with more than 85% of these due to heart attacks and strokes. Multiple peer-reviewed studies suggest that addressing these blockages in the coronary microcirculation could reduce this global burden.
“Real-time catheter-based MVO detection can provide healthcare professionals with valuable information about the extent and severity of the heart attack, which can inform immediate treatment decisions to impact patient outcomes," CorFlow CEO Paul Mead stated. “The CoFI System can be a game-changer as it is the first of its kind and can potentially revolutionize the field, which is ripe for innovation that makes a difference. The ‘next big thing’ in interventional cardiology should be driven by where the largest positive impact in outcomes can be made, and it is hard to imagine a clearer target in this regard than STEMI patients with MVO, the first indication we pursued. MOCA I shows that we can, and will, do better for these patients.”
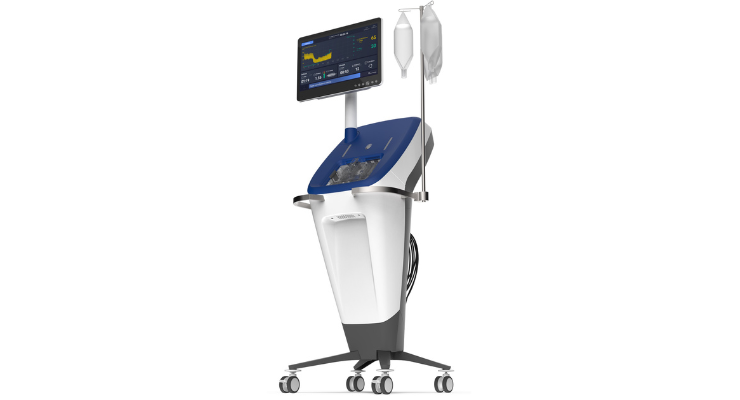
The Controlled Flow Infusion (CoFI) System is designed for diagnostic assessment of the coronary microcirculation in the cath lab, and to be a platform for controlled infusion of physician-specified diagnostic and therapeutic agents into the microcirculation. The CoFI System uses proprietary software algorithms to detect microvascular obstruction (MVO) and potentially other conditions related to microvascular function. The system the company plans to bring to the market is comprised of a next generation console, an advanced proprietary catheter with a pressure sensor wire, and single use infusion cassette. In addition to the primary clinical target addressing heart attack patients, the company intends to pursue other indications in both coronary and broader vascular use cases with the technology which are covered by broad patent families.
CorFlow Therapeutics AG, a venture-backed company headquartered in Switzerland, is advancing medical technology and cardiology to improve clinical outcomes for people with heart attacks and coronary artery disease. Two of the founders of the company are renowned interventional cardiologists, Drs. Rob Schwartz and Martin Rothman, who discovered acute unmet needs over decades of clinical practive. The company is currently raising a Series B funding round and is continuing to enroll patients in the ongoing European MOCA I clinical trial of the CoFI System, which will eventually be used for U.S. study submissions for clinical trials and commercialization of its technology.
The CoFI-System is not approved in the United States. It is an investigational device limited by U.S. federal law to investigational use and is not available for commercial distribution.
The company's MOCA I (Microvascular Obstruction with CoFI System Assessment) study is evaluating the safety and feasibility of the real-time catheter-based system to detect coronary microvascular obstruction MVO immediately after primary percutaneous coronary intervention (PPCI) and deliver timely therapeutic intracoronary agents. The trial data set included 30 patients with an ST elevation myocardial infarction (STEMI), the most severe type of heart attack.
“The data presented from the MOCA I study with the CorFlow system is very encouraging for our community of cardiologists, who are increasingly recognizing MVO and microvascular disease as a next frontier to improve outcomes in our field,” stated Dr. Azeem Latib of Montefiore Medical Center, New York, N.Y. “This emerging area is in its early days, but we can agree that a significant step forward is made by being able to diagnose these patients in the cath lab. With such a technology, we can imagine that many concomitant therapy options can soon be evaluated, since it is now clear that treating only the larger vessels is not enough.”
The system outputs a novel pressure parameter (PCoFI) for detection of MVO, which was evaluated for correlation with cardiac MRI measured MVO, considered the reference standard. For the patients who had the CoFI diagnostic sequence, the essential results of the study showed that the system provided a 95% sensitivity, 91% specificity, and 0.94 AUC for the ability to detect MVO measured by cardiac MRI. The CoFI device and procedure were deemed safe with no device-related deaths, flow-limiting dissections, or thrombotic events at 30 days, as adjudicated by an independent Clinical Events Committee.
The MOCA I study suggests that it might now be possible to have accurate, early detection which can lead to targeted treatment for these high-risk patients.
Current interventional cardiology technologies, addressing the larger coronary arteries, have been instrumental in saving patients from death and complications after a heart attack, reducing short term mortality over the last several decades. Yet for many patients, longer term outcomes are still poor despite current standard of care treatment. MVO, which is found to be in more than 50% of heart attack patients, has been shown to be the single most powerful prognostic indicator for negative outcomes for these patients, including heart failure and death. Heart disease remains the leading cause of death worldwide, with more than 85% of these due to heart attacks and strokes. Multiple peer-reviewed studies suggest that addressing these blockages in the coronary microcirculation could reduce this global burden.
“Real-time catheter-based MVO detection can provide healthcare professionals with valuable information about the extent and severity of the heart attack, which can inform immediate treatment decisions to impact patient outcomes," CorFlow CEO Paul Mead stated. “The CoFI System can be a game-changer as it is the first of its kind and can potentially revolutionize the field, which is ripe for innovation that makes a difference. The ‘next big thing’ in interventional cardiology should be driven by where the largest positive impact in outcomes can be made, and it is hard to imagine a clearer target in this regard than STEMI patients with MVO, the first indication we pursued. MOCA I shows that we can, and will, do better for these patients.”
The Controlled Flow Infusion (CoFI) System is designed for diagnostic assessment of the coronary microcirculation in the cath lab, and to be a platform for controlled infusion of physician-specified diagnostic and therapeutic agents into the microcirculation. The CoFI System uses proprietary software algorithms to detect microvascular obstruction (MVO) and potentially other conditions related to microvascular function. The system the company plans to bring to the market is comprised of a next generation console, an advanced proprietary catheter with a pressure sensor wire, and single use infusion cassette. In addition to the primary clinical target addressing heart attack patients, the company intends to pursue other indications in both coronary and broader vascular use cases with the technology which are covered by broad patent families.
CorFlow Therapeutics AG, a venture-backed company headquartered in Switzerland, is advancing medical technology and cardiology to improve clinical outcomes for people with heart attacks and coronary artery disease. Two of the founders of the company are renowned interventional cardiologists, Drs. Rob Schwartz and Martin Rothman, who discovered acute unmet needs over decades of clinical practive. The company is currently raising a Series B funding round and is continuing to enroll patients in the ongoing European MOCA I clinical trial of the CoFI System, which will eventually be used for U.S. study submissions for clinical trials and commercialization of its technology.
The CoFI-System is not approved in the United States. It is an investigational device limited by U.S. federal law to investigational use and is not available for commercial distribution.