Globe Newswire10.25.19
Stereotaxis, a developer of robotic technologies for the treatment of cardiac arrhythmias, has appointed Kimberly Peery as chief financial officer (CFO), effective Oct.1. Martin Stammer, who has served in the position since 2013, has accepted a job as CFO of a large professional services firm headquartered in the St. Louis area and resigned from the company.
“I have really enjoyed my time at Stereotaxis and firmly believe in the future of the company,” said Stammer. “I have the utmost confidence in Kim and her team and look forward to seeing them achieve many additional milestones.”
Peery joined Stereotaxis in 2003 and served as vice president of Finance. Prior to Stereotaxis, she served as a controller at various private companies. Peery is a certified public accountant and earned a bachelor of science degree in accounting and master of science degree in accounting and computer science from Southern Illinois University, Edwardsville.
“I appreciate the confidence placed in me by the Stereotaxis Board of Directors and look forward to becoming CFO in this exciting period for Stereotaxis,” Peery said.
David Fischel, chairman and CEO of Stereotaxis, commented, “I appreciate Marty’s contributions to Stereotaxis over many years and wish him well in his future endeavors. He leaves us in a strong position following our recently completed financing and uplisting to the NYSE American. I am excited to work with Kim and congratulate her on her new leadership role.”
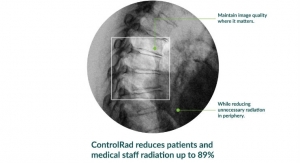
Stereotaxis develops robotic technologies designed to enhance the treatment of arrhythmias and perform endovascular procedures. Its mission is the discovery, development, and delivery of robotic systems, instruments, and information solutions for the interventional laboratory. These innovations help physicians provide patient care with robotic precision and safety, improved lab efficiency and productivity, and enhanced integration of procedural information. Stereotaxis’ robotic technology has received various regulatory clearances in the United States, European Union, Japan, Canada, China, and elsewhere. The Stereotaxis Genesis RMN System is CE marked and will become available in other global geographies subject to regulatory approvals. Stereotaxis Imaging Model S is CE marked and cleared by the U.S. Food and Drug Administration.
“I have really enjoyed my time at Stereotaxis and firmly believe in the future of the company,” said Stammer. “I have the utmost confidence in Kim and her team and look forward to seeing them achieve many additional milestones.”
Peery joined Stereotaxis in 2003 and served as vice president of Finance. Prior to Stereotaxis, she served as a controller at various private companies. Peery is a certified public accountant and earned a bachelor of science degree in accounting and master of science degree in accounting and computer science from Southern Illinois University, Edwardsville.
“I appreciate the confidence placed in me by the Stereotaxis Board of Directors and look forward to becoming CFO in this exciting period for Stereotaxis,” Peery said.
David Fischel, chairman and CEO of Stereotaxis, commented, “I appreciate Marty’s contributions to Stereotaxis over many years and wish him well in his future endeavors. He leaves us in a strong position following our recently completed financing and uplisting to the NYSE American. I am excited to work with Kim and congratulate her on her new leadership role.”
Stereotaxis develops robotic technologies designed to enhance the treatment of arrhythmias and perform endovascular procedures. Its mission is the discovery, development, and delivery of robotic systems, instruments, and information solutions for the interventional laboratory. These innovations help physicians provide patient care with robotic precision and safety, improved lab efficiency and productivity, and enhanced integration of procedural information. Stereotaxis’ robotic technology has received various regulatory clearances in the United States, European Union, Japan, Canada, China, and elsewhere. The Stereotaxis Genesis RMN System is CE marked and will become available in other global geographies subject to regulatory approvals. Stereotaxis Imaging Model S is CE marked and cleared by the U.S. Food and Drug Administration.