Business Wire12.18.17
Cagent Vascular, a developer of next-generation technology for vessel dilatation in cardiovascular disease interventions, has received CE Marking for the Serranator PTA Serration Balloon Catheter. Cagent Vascular has also achieved its ISO 13485 certification. The Serranator is one of a family of peripheral artery disease (PAD) technologies which incorporates proprietary Serration technology to an angioplasty balloon.
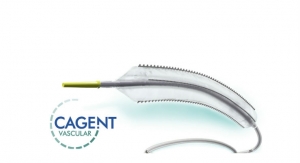
The Serranator has four embedded metal strips on an angioplasty balloon designed to create multiple longitudinal lines of interrupted micro-serrations to aid arterial expansion. Material that is serrated is more responsive to directed energy. In angioplasty, the serrated lines are more responsive to the balloon’s energy, thus achieving lumen expansion in a controlled and predictable way.
“Angioplasty is simple and inexpensive but doesn’t work very well. A distinct and unique advantage of the Serranator is that it capitalizes on the simplicity and familiarity of angioplasty while introducing a new and more effective method of vessel expansion,” Chief Medical Officer and co-founder Dr. Peter Schneider stated.
“Achieving CE Marking is an important milestone as we build upon physician enthusiasm for our device and finalize plans for successful commercialization,” CEO Carol Burns said. “In addition to the Serranator Alto device for use in treating diseased SFA and popliteal arteries, we are developing the Serranator Bass for the infrapopliteal arteries. Both devices are expected to be part of the commercial product offering in 2018. We believe both devices have the potential to be best-in-class devices.”
At this years’ TCT Conference, Dr. William Gray presented the PRELUDE study results. The study (n=25) assessed the Serranator Alto device in the superficial femoral and popliteal arteries. The subject population included 28 percent with a Chronic Total Occlusion (CTO). In 56 percent there was moderate to severe calcification as determined by the Yale Angiographic Core Laboratory. The acute results showed 100 percent device technical success and improvement in lumen gain from an average diameter stenosis of 88 percent to 23 percent after Serranator use. The bailout stent rate was 4 percent. There were no flow limiting dissections nor major adverse events. A subset of subjects (n=10) had Optical Coherence Tomography (OCT) or Intravascular Ultrasound (IVUS) imaging performed after Serranator use. A serration effect was shown in all 10 subjects. “The study showed safe and effective treatment in critical femoro-popliteal lesions with a low bailout stent rate and low final residual stenosis,” said Gray.
Cagent Vascular received U.S. Food and Drug Administration clearance for its first product, Serranator Alto, early this year. It is indicated for dilatation of lesions in the iliac, femoral, iliofemoral, and popliteal arteries and for the treatment of obstructive lesions of native or synthetic arteriovenous dialysis fistulae.
Cagent Vascular develops technology for vessel dilatation. It is the second cardiovascular company formed by co-founders Burns, Schneider (a vascular surgeon), and Robert Giasolli, vice president of Research and Development. The company is based in Wayne, Pa.
The Serranator has four embedded metal strips on an angioplasty balloon designed to create multiple longitudinal lines of interrupted micro-serrations to aid arterial expansion. Material that is serrated is more responsive to directed energy. In angioplasty, the serrated lines are more responsive to the balloon’s energy, thus achieving lumen expansion in a controlled and predictable way.
“Angioplasty is simple and inexpensive but doesn’t work very well. A distinct and unique advantage of the Serranator is that it capitalizes on the simplicity and familiarity of angioplasty while introducing a new and more effective method of vessel expansion,” Chief Medical Officer and co-founder Dr. Peter Schneider stated.
“Achieving CE Marking is an important milestone as we build upon physician enthusiasm for our device and finalize plans for successful commercialization,” CEO Carol Burns said. “In addition to the Serranator Alto device for use in treating diseased SFA and popliteal arteries, we are developing the Serranator Bass for the infrapopliteal arteries. Both devices are expected to be part of the commercial product offering in 2018. We believe both devices have the potential to be best-in-class devices.”
At this years’ TCT Conference, Dr. William Gray presented the PRELUDE study results. The study (n=25) assessed the Serranator Alto device in the superficial femoral and popliteal arteries. The subject population included 28 percent with a Chronic Total Occlusion (CTO). In 56 percent there was moderate to severe calcification as determined by the Yale Angiographic Core Laboratory. The acute results showed 100 percent device technical success and improvement in lumen gain from an average diameter stenosis of 88 percent to 23 percent after Serranator use. The bailout stent rate was 4 percent. There were no flow limiting dissections nor major adverse events. A subset of subjects (n=10) had Optical Coherence Tomography (OCT) or Intravascular Ultrasound (IVUS) imaging performed after Serranator use. A serration effect was shown in all 10 subjects. “The study showed safe and effective treatment in critical femoro-popliteal lesions with a low bailout stent rate and low final residual stenosis,” said Gray.
Cagent Vascular received U.S. Food and Drug Administration clearance for its first product, Serranator Alto, early this year. It is indicated for dilatation of lesions in the iliac, femoral, iliofemoral, and popliteal arteries and for the treatment of obstructive lesions of native or synthetic arteriovenous dialysis fistulae.
Cagent Vascular develops technology for vessel dilatation. It is the second cardiovascular company formed by co-founders Burns, Schneider (a vascular surgeon), and Robert Giasolli, vice president of Research and Development. The company is based in Wayne, Pa.