Toshiba America Medical Systems Inc. 12.08.16
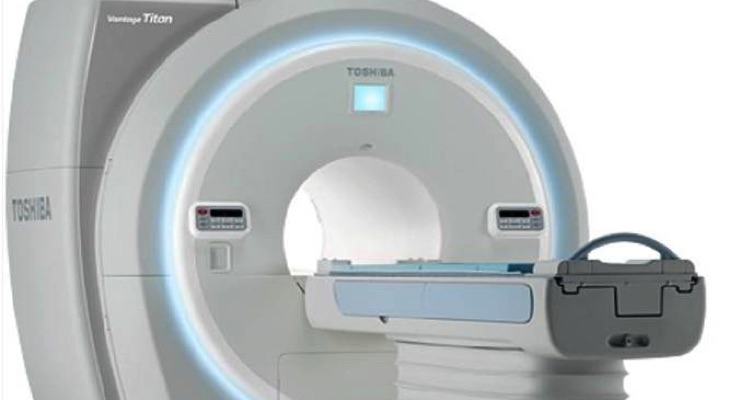
Presenting significant improvements to cardiac magnetic resonane (MR) imaging, Toshiba America Medical System Inc.’s Vantage Titan 1.5T/cS Edition with M-Power V3.6 software has received U.S. Food and Drug Administration clearance. The new system retains all the features of the Vantage Titan 1.5T MR with added technology to simplify complex cardiac exams.
The Vantage Titan/cS Edition offers cardiologists and radiologists a comprehensive suite of innovative applications to help them improve their workflow and patient comfort:
“The Vantage Titan/cS Edition gives healthcare providers the ability to diversify their MR imaging offerings and reduce their exam times, creating a new opportunity for routine cardiac MR,” said Dominic Smith, senior director, CT, PET/CT, and MR Business Units at Toshiba America Medical Systems. “Toshiba Medical understands the patient experience and business needs of MR, and the Vantage Titan/cS Edition offers customers innovative MR imaging solutions that meet a variety of clinical needs, without compromising patient satisfaction.”
With headquarters in Tustin, Calif., Toshiba America Medical Systems markets, sells, distributes and services radiology and cardiovascular systems, including CT, MR, ultrasound, X-ray and cardiovascular equipment, and coordinates clinical diagnostic imaging research for all modalities in the United States.
The Vantage Titan/cS Edition offers cardiologists and radiologists a comprehensive suite of innovative applications to help them improve their workflow and patient comfort:
- SUREVOI with CardioLine+ (Option): The system offers SUREVOI Cardiac anatomy recognition technology to automatically determine the position of the heart. The enhanced CardioLine application CardioLine+ works with SUREVOI to automatically detect anatomical landmarks for accurate and reproducible positioning to complete alignment of 14 standard cardiac views with minimal operator interaction.
- Multi b-Value Diffusion: The Vantage Titan/cS Edition allows acquisition of multiple b-values in a single scan enabling physicians to quickly identify tissue diffusivity.
- Multi-echo T2 Mapping: The system also supports Multi-echo T2 Mapping, enabling physicians to visualize T2 map cartilage images to aid them in making a diagnosis and determining if treatment is needed.
- Vitrea Extend (Option): A state-of-the-art post-processing application, Vitrea Extend permits clinicians to complete post-processing at a separate workstation while allowing the next patient exam to be conducted at the MR system. The Vitrea Extend solution comes with expert packages powered by the latest post-processing applications made by Olea Medical and Medis.
“The Vantage Titan/cS Edition gives healthcare providers the ability to diversify their MR imaging offerings and reduce their exam times, creating a new opportunity for routine cardiac MR,” said Dominic Smith, senior director, CT, PET/CT, and MR Business Units at Toshiba America Medical Systems. “Toshiba Medical understands the patient experience and business needs of MR, and the Vantage Titan/cS Edition offers customers innovative MR imaging solutions that meet a variety of clinical needs, without compromising patient satisfaction.”
With headquarters in Tustin, Calif., Toshiba America Medical Systems markets, sells, distributes and services radiology and cardiovascular systems, including CT, MR, ultrasound, X-ray and cardiovascular equipment, and coordinates clinical diagnostic imaging research for all modalities in the United States.