PR Newswire11.01.16
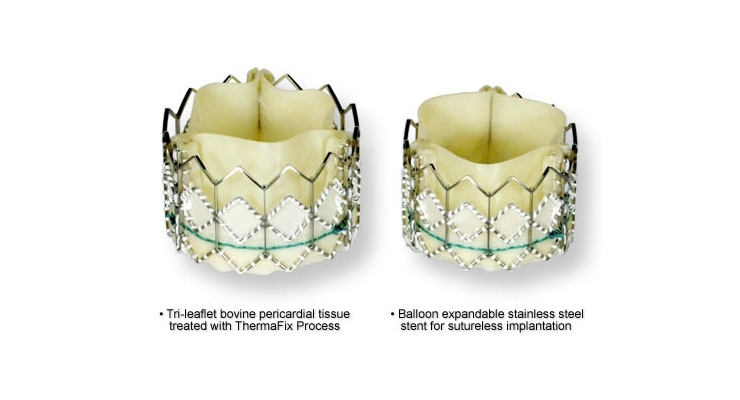
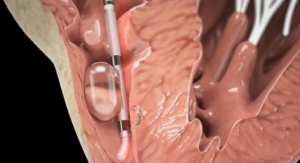
Edwards Lifesciences Corporation, developer of patient-focused innovations for structural heart disease and critical care monitoring, today announced new 5-year hemodynamic data from the PARTNER Trial demonstrating excellent durability of the first-generation Edwards SAPIEN transcatheter heart valve. This evaluation of 2,404 patients, encompassing 10,560 echoes and 6,493 patient years of follow-up, is the largest core lab based study of transcatheter heart valves to date. Results were presented during the late-breaking clinical trials session at the 28th Transcatheter Cardiovascular Therapeutics (TCT), the annual scientific symposium of the Cardiovascular Research Foundation, in Washington, D.C.
"In the largest, most robust study of its kind, population hemodynamic trends show excellent durability of the SAPIEN transcatheter heart valve over five years, with virtually no structural valve deterioration," said Pamela Douglas, M.D., Ursula Geller professor of research in cardiovascular disease at Duke University and director of the Imaging Program at Duke Clinical Research Institute, who presented the data. "Similarly, large adverse hemodynamic echo findings in individual patients are rare in this protocol-driven database. Together, these data demonstrate excellent mid-term durability of THV, suggesting that the low 5-year survival observed in this cohort is not due to adverse hemodynamics."
Using three standard echocardiographic parameters, the study assessed hemodynamics (at 7 days, 30 days, 6 months, 1 year, 2 years, 3 years, 4 years, and 5 years post-implant) and related the findings to clinical events of death and aortic valve re-intervention.
"We are very pleased with the scientifically rigorous 5-year outcomes data presented at TCT," said Larry L. Wood, Edwards' corporate vice president, transcatheter heart valves. "While this research is meaningful and important information for both clinicians and patients, we see this as the starting point for further evaluations, and our intention is to use the PARTNER and PARTNER II Trial data to generate additional longer-term evidence on durability for the SAPIEN family of heart valves."
The PARTNER Trial was the world's first prospective, randomized and controlled trial for transcatheter heart valves.
"In the largest, most robust study of its kind, population hemodynamic trends show excellent durability of the SAPIEN transcatheter heart valve over five years, with virtually no structural valve deterioration," said Pamela Douglas, M.D., Ursula Geller professor of research in cardiovascular disease at Duke University and director of the Imaging Program at Duke Clinical Research Institute, who presented the data. "Similarly, large adverse hemodynamic echo findings in individual patients are rare in this protocol-driven database. Together, these data demonstrate excellent mid-term durability of THV, suggesting that the low 5-year survival observed in this cohort is not due to adverse hemodynamics."
Using three standard echocardiographic parameters, the study assessed hemodynamics (at 7 days, 30 days, 6 months, 1 year, 2 years, 3 years, 4 years, and 5 years post-implant) and related the findings to clinical events of death and aortic valve re-intervention.
"We are very pleased with the scientifically rigorous 5-year outcomes data presented at TCT," said Larry L. Wood, Edwards' corporate vice president, transcatheter heart valves. "While this research is meaningful and important information for both clinicians and patients, we see this as the starting point for further evaluations, and our intention is to use the PARTNER and PARTNER II Trial data to generate additional longer-term evidence on durability for the SAPIEN family of heart valves."
The PARTNER Trial was the world's first prospective, randomized and controlled trial for transcatheter heart valves.