Business Wire06.30.16
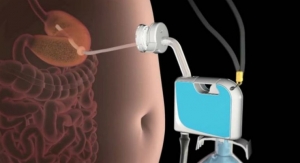
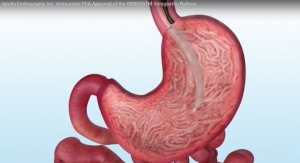
Apollo Endosurgery, Inc., a leader in minimally invasive endoscopic surgical products for bariatric and gastrointestinal procedures, today announced the start of its U.S. post-approval study of the ORBERA Intragastric Balloon, a medical device used to assist adult patients suffering from obesity—with a body mass index (BMI) of 30 to 40—in losing and maintaining weight.
The first patient in the Post-Approval Study Evaluating the Long-Term Safety and Effectiveness of ORBERA was enrolled earlier this month in New Orleans, Louisiana, by the site’s lead investigator, bariatric surgeon Dr. Rachel Moore.
“ORBERA’s post-approval clinical study exemplifies Apollo’s commitment to offering safe and effective weight loss solutions to patients and their physicians in the United States,” Dr. Moore said. “The resulting data will help clinicians worldwide improve the treatment of obesity with a proven, minimally invasive option.”
The post-approval study is a prospective, multicenter, open-label study designed to demonstrate the safety and effectiveness of ORBERA as an adjunct to weight reduction for obese adults (22 years of age and older) with a Body Mass Index (BMI) of ≥ 30 kg/m2 and BMI ≤ 40 kg/m2. The study will involve approximately 255 subjects at up to 20 U.S. sites.
The U.S. pivotal ORBERA clinical trial first announced in May of 2015 was a multicenter, prospective, randomized, non-blinded comparative study where a total of 255 patients suffering from obesity with a BMI between 30 and 40 were randomized to treatment or control in a 1:1 ratio. The treatment group (125 patients) underwent placement of the ORBERA balloon followed by removal after six months. They concurrently participated in a 12-month behavioral modification program. The control group (130 patients) participated in the 12-month behavioral modification program alone. For patients in the treatment group, the device was removed at month six, with regular office visits continuing through one year.
Detailed findings from the trial include:
“Clinical confidence in medical devices comes from clinical evidence. Since ORBERA’s inception, there have been over 230 peer-reviewed ORBERA publications around the globe reporting on data of over 8,000 patients,” said Todd Newton, Chief Executive Officer of Apollo Endosurgery. “Retrospective and pivotal studies show, time and again, that ORBERA is safe and effective, and helps patients lose the weight and also keep the weight off even after the device is removed. We believe the U.S. post-approval study will add to this unmatched dataset.”
The first patient in the Post-Approval Study Evaluating the Long-Term Safety and Effectiveness of ORBERA was enrolled earlier this month in New Orleans, Louisiana, by the site’s lead investigator, bariatric surgeon Dr. Rachel Moore.
“ORBERA’s post-approval clinical study exemplifies Apollo’s commitment to offering safe and effective weight loss solutions to patients and their physicians in the United States,” Dr. Moore said. “The resulting data will help clinicians worldwide improve the treatment of obesity with a proven, minimally invasive option.”
The post-approval study is a prospective, multicenter, open-label study designed to demonstrate the safety and effectiveness of ORBERA as an adjunct to weight reduction for obese adults (22 years of age and older) with a Body Mass Index (BMI) of ≥ 30 kg/m2 and BMI ≤ 40 kg/m2. The study will involve approximately 255 subjects at up to 20 U.S. sites.
The U.S. pivotal ORBERA clinical trial first announced in May of 2015 was a multicenter, prospective, randomized, non-blinded comparative study where a total of 255 patients suffering from obesity with a BMI between 30 and 40 were randomized to treatment or control in a 1:1 ratio. The treatment group (125 patients) underwent placement of the ORBERA balloon followed by removal after six months. They concurrently participated in a 12-month behavioral modification program. The control group (130 patients) participated in the 12-month behavioral modification program alone. For patients in the treatment group, the device was removed at month six, with regular office visits continuing through one year.
Detailed findings from the trial include:
- At month six, the ORBERA group achieved a mean of 38.4 percent Excess Weight Loss (EWL).
- Mean Total Body Weight Loss (TBWL) at six months was 10.2 percent for the treatment group compared to 3.3 percent TBWL for the control group.
- The ORBERA group lost 3.1 times as much weight as the control group at six months.
- The ORBERA group also lost significantly more weight than the control group over the course of the study, and was able to maintain significant weight loss through month 12, six months after removal of the device.
“Clinical confidence in medical devices comes from clinical evidence. Since ORBERA’s inception, there have been over 230 peer-reviewed ORBERA publications around the globe reporting on data of over 8,000 patients,” said Todd Newton, Chief Executive Officer of Apollo Endosurgery. “Retrospective and pivotal studies show, time and again, that ORBERA is safe and effective, and helps patients lose the weight and also keep the weight off even after the device is removed. We believe the U.S. post-approval study will add to this unmatched dataset.”