10.12.15
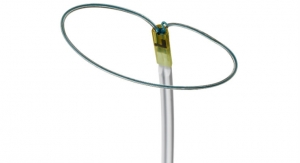
Breathe Technologies Inc. has secured U.S. Food and Drug Administration 510(k) clearance of its Life2000 ventilation system.
The system is indicated for continuous or intermittent ventilatory support in adult patients who require invasive (through an ET tube) or noninvasive (through a mask) positive pressure ventilation. The system can be used in home and hospital settings, company executives said in a news release.
The devicemaker plans to launch the product in the U.S. late this year.
Breathe Technologies creates products for patients with respiratory insufficiencies and neuromuscular diseases.
"We are pleased to announce the FDA clearance of the Breathe Technologies Life2000 Ventilation system," said Larry Mastrovich, president/CEO of Breathe Technologies. "This clearance underscores our commitment to providing healthcare providers and patients with another therapeutic option for critical care ventilation."
Based in Irvine, Calif., Breathe Technologies develops and manufactures technology for patients with respiratory insufficiency and neuromuscular diseases. The company also manufactures the Non-Invasive Open Ventilation (NIOV) system, a lightweight and wearable ventilator that reduces the work of breathing and supports improved patient mobility. In two clinical trials, NIOV system demonstrated improvements in six-minute walk tests (6MWT) in subjects with severe chronic obstructive pulmonary disease and other forms of respiratory insufficiency.
In the first study, conducted at two pulmonary rehabilitation centers, NIOV increased 6MWT distance in 25 of 30 subjects compared to standard oxygen therapy via nasal cannula.In a subset of subjects in the second study, with baseline 6MWT distances of less than 300 meters, the mean improvement was 73 meters.In both clinical studies, the NIOV system was well-tolerated and subjects reported that it was comfortable to wear.
Breathe Technologies' investors include Kleiner Perkins Caufield & Byers, Deerfield Management Company LP, Roddenbury Foundation, DAG Ventures, Morgan Creek, BioMed Ventures, and Synergy Partners International.
The system is indicated for continuous or intermittent ventilatory support in adult patients who require invasive (through an ET tube) or noninvasive (through a mask) positive pressure ventilation. The system can be used in home and hospital settings, company executives said in a news release.
The devicemaker plans to launch the product in the U.S. late this year.
Breathe Technologies creates products for patients with respiratory insufficiencies and neuromuscular diseases.
"We are pleased to announce the FDA clearance of the Breathe Technologies Life2000 Ventilation system," said Larry Mastrovich, president/CEO of Breathe Technologies. "This clearance underscores our commitment to providing healthcare providers and patients with another therapeutic option for critical care ventilation."
Based in Irvine, Calif., Breathe Technologies develops and manufactures technology for patients with respiratory insufficiency and neuromuscular diseases. The company also manufactures the Non-Invasive Open Ventilation (NIOV) system, a lightweight and wearable ventilator that reduces the work of breathing and supports improved patient mobility. In two clinical trials, NIOV system demonstrated improvements in six-minute walk tests (6MWT) in subjects with severe chronic obstructive pulmonary disease and other forms of respiratory insufficiency.
In the first study, conducted at two pulmonary rehabilitation centers, NIOV increased 6MWT distance in 25 of 30 subjects compared to standard oxygen therapy via nasal cannula.In a subset of subjects in the second study, with baseline 6MWT distances of less than 300 meters, the mean improvement was 73 meters.In both clinical studies, the NIOV system was well-tolerated and subjects reported that it was comfortable to wear.
Breathe Technologies' investors include Kleiner Perkins Caufield & Byers, Deerfield Management Company LP, Roddenbury Foundation, DAG Ventures, Morgan Creek, BioMed Ventures, and Synergy Partners International.