08.27.15
Pulmonx now has more clinical evidence to support the safety and efficacy of its Zephyr Endobronchial Valve (EBV) in severe emphysema patients.
The company recently shared six-month results from the independent, randomized, controlled STELVIO trial, which compared the Zephyr EBV with standard medical care in 84 severe emphysema patients. Using the Chartis system, 68 patients were confirmed to have no collateral ventilation, independent of disease distribution, and were treated with the Zephyr EBV. At six months, statistically and clinically significant improvements in pulmonary function, exercise capacity and quality of life were seen in this patient cohort, as described in the following chart:
The primary investigator of the STELVIO trial is Dirk-Jan Slebos, M.D., of the Department of Pulmonary Diseases and the Groningen Research Institute for Asthma and COPD, University Medical Center Groningen in The Netherlands. The trial has been funded by a Dutch government grant from ZonMW and the University Medical Center Groningen in The Netherlands.
“The benefit of the Zephyr EBV is undeniable in emphysema patients without collateral ventilation,” said Slebos. “What is clear is that we, as physicians, must both carefully select the patients for this treatment and technically perform a successful procedure. When we do, the improvements can be life-changing for a patient group that up until now has had very limited treatment options.”
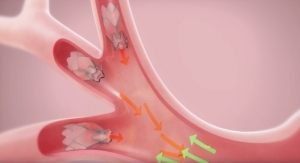
The Zephyr EBV is a minimally invasive treatment for severe emphysema that has been proven in over a decade of clinical experience to significantly improve the lung function, exercise tolerance and quality of life for patients receiving treatment, company executives said in a news release. A proprietary diagnostic – the Chartis System – is utilized prior to the procedure to identify likely responders. In the procedure, tiny, one-way valves are placed in the lungs to block airflow to diseased regions to achieve lung volume reduction. As a result, the remaining healthy regions may function more efficiently, enabling better breathing and an improved quality of life. Zephyr EBVs have been implanted globally in more than 10,000 patients.
“The STELVIO study provides independent confirmation that when patients are selected using the Chartis system, they have a high likelihood of achieving clinically meaningful improvement in lung function, exercise capacity and quality of life from Zephyr EBV therapy,” Pulmonx CEO Glen French said.
Three additional randomized controlled trials – VENT, EuroVENT and BeLieVeR-HIFi – have demonstrated the significant benefits of the Zephyr EBV in severe emphysema patients with heterogeneous disease, and a U.S. pivotal trial – the LIBERATE study – currently is enrolling.
Based in Redwood City, Calif., and Neuchâtel, Switzerland, Pulmonx is an interventional pulmonology company that develops lung disease treatment technologies.
The company recently shared six-month results from the independent, randomized, controlled STELVIO trial, which compared the Zephyr EBV with standard medical care in 84 severe emphysema patients. Using the Chartis system, 68 patients were confirmed to have no collateral ventilation, independent of disease distribution, and were treated with the Zephyr EBV. At six months, statistically and clinically significant improvements in pulmonary function, exercise capacity and quality of life were seen in this patient cohort, as described in the following chart:
| PATIENTS TREATED WITH ZEPHYR EBV | ||||
| Measurement (MCID) | Improvement from Control | Responder Rate | ||
| FEV1 percent (15%) | 23% | 63% | ||
| 6MWT (26 m) | 106 meters | 87% | ||
| SGRQ (-4 points) | -15 points | 79% | ||
The primary investigator of the STELVIO trial is Dirk-Jan Slebos, M.D., of the Department of Pulmonary Diseases and the Groningen Research Institute for Asthma and COPD, University Medical Center Groningen in The Netherlands. The trial has been funded by a Dutch government grant from ZonMW and the University Medical Center Groningen in The Netherlands.
“The benefit of the Zephyr EBV is undeniable in emphysema patients without collateral ventilation,” said Slebos. “What is clear is that we, as physicians, must both carefully select the patients for this treatment and technically perform a successful procedure. When we do, the improvements can be life-changing for a patient group that up until now has had very limited treatment options.”
The Zephyr EBV is a minimally invasive treatment for severe emphysema that has been proven in over a decade of clinical experience to significantly improve the lung function, exercise tolerance and quality of life for patients receiving treatment, company executives said in a news release. A proprietary diagnostic – the Chartis System – is utilized prior to the procedure to identify likely responders. In the procedure, tiny, one-way valves are placed in the lungs to block airflow to diseased regions to achieve lung volume reduction. As a result, the remaining healthy regions may function more efficiently, enabling better breathing and an improved quality of life. Zephyr EBVs have been implanted globally in more than 10,000 patients.
“The STELVIO study provides independent confirmation that when patients are selected using the Chartis system, they have a high likelihood of achieving clinically meaningful improvement in lung function, exercise capacity and quality of life from Zephyr EBV therapy,” Pulmonx CEO Glen French said.
Three additional randomized controlled trials – VENT, EuroVENT and BeLieVeR-HIFi – have demonstrated the significant benefits of the Zephyr EBV in severe emphysema patients with heterogeneous disease, and a U.S. pivotal trial – the LIBERATE study – currently is enrolling.
Based in Redwood City, Calif., and Neuchâtel, Switzerland, Pulmonx is an interventional pulmonology company that develops lung disease treatment technologies.