08.06.15
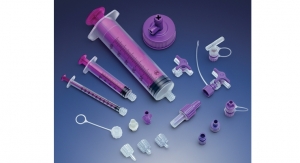
Officials with Bi-Link, a global contract manufacturer headquartered in Bloomingdale, Ill., report that the company’s Bloomingdale and Juarez, Mexico, facilities have been certified for ISO 13485:2003.
Joining Bi-Link’s manufacturing facility in Shanghai, China, the certification of compliance recognizes Bi-Link’s policies, practices and procedures are able to consistently meet customer and regulatory requirements for medical device manufacturing and other related services.
“Although Bi-Link has been serving the healthcare and medical device industries for over a decade, certification of its North American manufacturing facilities demonstrates Bi-Link’s dedication to delivering a consistent level of quality, efficiency and responsiveness across the globe,” said Terri Marion, medical segment marketing manager of Bi-Link. “Many of our current customers are global, industry-leading OEMs who must meet increasingly strict technical, functional and regulatory requirements. Being ISO 13485-certified ensures our customers that we are committed to supporting them and their needs.”
Bi-Link has more than 50 years of experience helping product engineers solve complex design challenges with a process the company calls “Mind to Market”— a vertically integrated process that officials claim lowers overall life-cycle cost and accelerates time to market by helping engineers validate designs based on solid DFM principles and rapid “pre-prototypes.”
Bi-Link was founded in 1961 and provides rapid prototyping, in-house tooling, precision metal stamping, plastic injection and insert molding, and automated assembly. Bi-link collaborates with healthcare and medical device engineering teams to solve complex design challenges from early stages through full-scale manufacturing.
Joining Bi-Link’s manufacturing facility in Shanghai, China, the certification of compliance recognizes Bi-Link’s policies, practices and procedures are able to consistently meet customer and regulatory requirements for medical device manufacturing and other related services.
“Although Bi-Link has been serving the healthcare and medical device industries for over a decade, certification of its North American manufacturing facilities demonstrates Bi-Link’s dedication to delivering a consistent level of quality, efficiency and responsiveness across the globe,” said Terri Marion, medical segment marketing manager of Bi-Link. “Many of our current customers are global, industry-leading OEMs who must meet increasingly strict technical, functional and regulatory requirements. Being ISO 13485-certified ensures our customers that we are committed to supporting them and their needs.”
Bi-Link has more than 50 years of experience helping product engineers solve complex design challenges with a process the company calls “Mind to Market”— a vertically integrated process that officials claim lowers overall life-cycle cost and accelerates time to market by helping engineers validate designs based on solid DFM principles and rapid “pre-prototypes.”
Bi-Link was founded in 1961 and provides rapid prototyping, in-house tooling, precision metal stamping, plastic injection and insert molding, and automated assembly. Bi-link collaborates with healthcare and medical device engineering teams to solve complex design challenges from early stages through full-scale manufacturing.