07.01.15
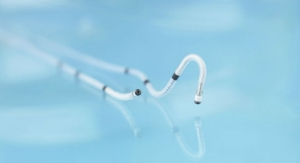
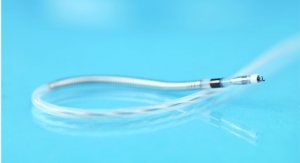
Biotronik SE & Co. KG has launched its the Pantera Pro semi-compliant coronary balloon dilatation catheter in Europe. The Pantera Pro balloon received CE mark earlier this year.
The first dilatation procedures with Pantera Pro were performed in 13 countries, based on which more than 700 product uses were evaluated.
"When treating patients with complex vascular anatomy, it can be difficult to ensure access to the lesion site and to achieve proper dilatation," said Juan F. Iglesias, M.D., of University Hospital in Lausanne, Switzerland. "Thanks to Pantera Pro's slim shoulder design and hydrophilic coating, even lesions affecting tight or challenging vessels may be more easily and reliably crossed."
Pantera Pro has been completely redesigned from the previous generation of Pantera balloons, integrating many progressive design and technology features to improve reach and enhance crossability. A new hydrophilic balloon coating and proprietary balloon material in the 1.25 millimeter to 2 millimeter size range ensures enhanced durability, according to Biontronik. In addition to these refinements to the smaller balloon sizes, the profile of the catheter's distal guide wire support section as measured in bench testing was reduced, thereby improving performance during kissing balloon technique and reducing friction within the vessel by up to 43 percent compared to competitors, company executives noted in a news release. Based on the first dilatation procedures, physicians have indicated that Pantera Pro performs better in 68 percent of cases than other semi-compliant balloons.
"The first users of Pantera Pro have discovered this workhorse balloon demonstrates excellent pushability and crossability, even in tight lesions," said Alexander Uhl, Ph.D., vice president of marketing for Biotronik's Vascular Intervention division. "Following in the footsteps of the Pantera LEO non-compliant and the Pantera Lux drug-coated balloons, the European launch of Pantera Pro establishes the Biotronik coronary balloon portfolio as the most versatile on the market."
Pantera Pro is touted by the company as a next-generation semi-compliant coronary "workhorse" balloon catheter designed to dilate stenotic segments in coronary arteries or bypass grafts. It is available in diameters from 1.25 millimeters to 4 millimeters and lengths from 6 millimeters to 30 millimeters.
Biotronik is based in Berlin, Germany, and has a global workforce of more than 5,600 employees. Its U.S. headquarters is located in Lake Oswego, Ore.
The first dilatation procedures with Pantera Pro were performed in 13 countries, based on which more than 700 product uses were evaluated.
"When treating patients with complex vascular anatomy, it can be difficult to ensure access to the lesion site and to achieve proper dilatation," said Juan F. Iglesias, M.D., of University Hospital in Lausanne, Switzerland. "Thanks to Pantera Pro's slim shoulder design and hydrophilic coating, even lesions affecting tight or challenging vessels may be more easily and reliably crossed."
Pantera Pro has been completely redesigned from the previous generation of Pantera balloons, integrating many progressive design and technology features to improve reach and enhance crossability. A new hydrophilic balloon coating and proprietary balloon material in the 1.25 millimeter to 2 millimeter size range ensures enhanced durability, according to Biontronik. In addition to these refinements to the smaller balloon sizes, the profile of the catheter's distal guide wire support section as measured in bench testing was reduced, thereby improving performance during kissing balloon technique and reducing friction within the vessel by up to 43 percent compared to competitors, company executives noted in a news release. Based on the first dilatation procedures, physicians have indicated that Pantera Pro performs better in 68 percent of cases than other semi-compliant balloons.
"The first users of Pantera Pro have discovered this workhorse balloon demonstrates excellent pushability and crossability, even in tight lesions," said Alexander Uhl, Ph.D., vice president of marketing for Biotronik's Vascular Intervention division. "Following in the footsteps of the Pantera LEO non-compliant and the Pantera Lux drug-coated balloons, the European launch of Pantera Pro establishes the Biotronik coronary balloon portfolio as the most versatile on the market."
Pantera Pro is touted by the company as a next-generation semi-compliant coronary "workhorse" balloon catheter designed to dilate stenotic segments in coronary arteries or bypass grafts. It is available in diameters from 1.25 millimeters to 4 millimeters and lengths from 6 millimeters to 30 millimeters.
Biotronik is based in Berlin, Germany, and has a global workforce of more than 5,600 employees. Its U.S. headquarters is located in Lake Oswego, Ore.