04.08.15
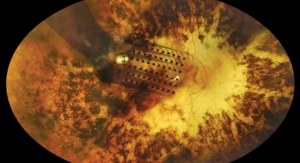
Second Sight Medical Products Inc., a developer of implantable visual prosthetics to restore functional vision to blind patients, today announced the first successful implant of a mechanical model of the Orion I Visual Cortical Prosthesis in an animal study.
The first implant was performed as part of a phase I pre-clinical study, which is designed to evaluate fit, form, stability and biocompatibility.
The study, which is expected to run through the end of the year, is the first major milestone in the development of the Orion I, company officials reported. Fully functional prototypes are expected to be completed later this year with active animal implants scheduled to begin by Q1 2016. The first human clinical trials are planned to begin by the first quarter of 2017. Assuming positive initial results in patients and discussions with regulators, an expanded pivotal clinical trial for global market approvals is planned.
"This is a major milestone not only for the Company but, more importantly, those affected by virtually all forms of blindness," said Robert Greenberg, M.D., Ph.D., president and CEO of Sylmar, Calif.-based Second Sight. "Following the success of Argus II in patients with Retinitis Pigmentosa, we are looking forward to extending the hope of restoring some useful vision to nearly all blind individuals with the Orion I."
The Orion I technology is based on the U.S. Food and Drug Administration-approved Argus II Retinal Prosthesis System, but with updates to the electrode neural interface—moving from the retina to the visual cortex. Implanted on the surface of the visual cortex located within the occipital lobe of the brain, Orion will bypass the retina and optic nerve altogether. This potentially offers hope for treating patients with nearly all forms of blindness where the optic nerve or retina is completely damaged, as in glaucoma, diabetic retinopathy, retinal detachments, trauma, infection, and others. The population of legally blind people potentially eligible for the Orion I is about 6 million worldwide, Second Sight officials predict.
The first implant was performed as part of a phase I pre-clinical study, which is designed to evaluate fit, form, stability and biocompatibility.
The study, which is expected to run through the end of the year, is the first major milestone in the development of the Orion I, company officials reported. Fully functional prototypes are expected to be completed later this year with active animal implants scheduled to begin by Q1 2016. The first human clinical trials are planned to begin by the first quarter of 2017. Assuming positive initial results in patients and discussions with regulators, an expanded pivotal clinical trial for global market approvals is planned.
"This is a major milestone not only for the Company but, more importantly, those affected by virtually all forms of blindness," said Robert Greenberg, M.D., Ph.D., president and CEO of Sylmar, Calif.-based Second Sight. "Following the success of Argus II in patients with Retinitis Pigmentosa, we are looking forward to extending the hope of restoring some useful vision to nearly all blind individuals with the Orion I."
The Orion I technology is based on the U.S. Food and Drug Administration-approved Argus II Retinal Prosthesis System, but with updates to the electrode neural interface—moving from the retina to the visual cortex. Implanted on the surface of the visual cortex located within the occipital lobe of the brain, Orion will bypass the retina and optic nerve altogether. This potentially offers hope for treating patients with nearly all forms of blindness where the optic nerve or retina is completely damaged, as in glaucoma, diabetic retinopathy, retinal detachments, trauma, infection, and others. The population of legally blind people potentially eligible for the Orion I is about 6 million worldwide, Second Sight officials predict.