02.19.15
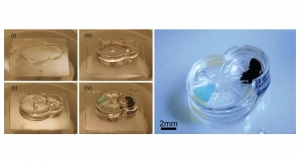
A new paper in the journal Tissue Engineering Part C: Methods outlines a new instrument for 3-D printing live tissue. The instrument is called the Bio-Pick, Place and Perfuse, or Bio-P3, and it picks up large complex multicellular building parts, transports them to a build area, and precisely places the parts at desired locations while perfusing the parts. According to the authors, these assembled parts can subsequently fuse to form a larger contiguous tissue construct.
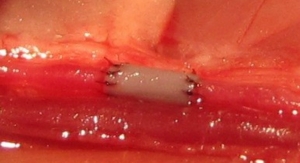
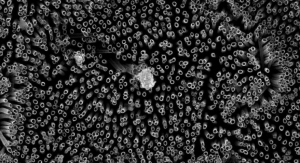
The research team formed multicellular microtissues by seeding cells into nonadhesive micro-molds, wherein cells self-assembled scaffold-free parts in various shapes. After removal from the molds, the parts were gripped, transported and released using the Bio-P3 with little to no effect on cell viability or part structure.
Scaffold-free building parts help to address several of the engineering and biological challenges to large tissue biofabrication, the researchers wrote. The Bio-P3 is designed to provide controlled gripping, placing, stacking and perfusing of living building parts for solid organ fabrication.
According to the authors, a “grand challenge” of tissue engineering has been to construct large living structures with high cell density akin to native organs and to sustain the viability of the structures while reproducing in vivo function. Scaffold-based tissue engineering methods usually involve either decellularizing a native organ and reseeding the residual extracellular matrix with the desired cells or seeding a biocompatible fabricated scaffold with cells. Scaffold-free methods, including pellet culture and hanging drops, are generally limited by the small scale of the produced tissue construct. Cell sheets, while larger in scale, have had only limited success in specific applications such as vascular tissue engineering.
The potential clinical impact of engineered tissues in treating disease is vast, the paper read. Liver failure, for example, leads to 1 million deaths per year worldwide, the 10th most common overall cause of death. This represents enormous direct and indirect healthcare costs. The supply of donor organs is far too small to meet the need, and many patients die while waiting on the transplant list. As the obesity epidemic increases, nonalcoholic fatty liver disease leading to liver failure will become an even more important clinical issue. So manufacturing live tissue by any means would count as a major breakthrough for a swath of applications.
The Bio-P3 builds on the principles of the pick and place devices used in the high-speed assembly of multicomponent electronics. The authors position the device as having exciting potential applications in solid organ tissue engineering as a platform for applications such as organ function replacement, solid tumor modeling, or three-dimensional drug testing.
“This device can be the long-expected breakthrough in the field of regenerative medicine and hopefully allow the fabrication of large 3-D organs and tissues," says John A. Jansen, DDS, Ph.D., co-editor-in-chief of Tissue Engineering, Part C: Methods and professor and head of dentistry, Radboud University Medical Center, The Netherlands.
The research team formed multicellular microtissues by seeding cells into nonadhesive micro-molds, wherein cells self-assembled scaffold-free parts in various shapes. After removal from the molds, the parts were gripped, transported and released using the Bio-P3 with little to no effect on cell viability or part structure.
Scaffold-free building parts help to address several of the engineering and biological challenges to large tissue biofabrication, the researchers wrote. The Bio-P3 is designed to provide controlled gripping, placing, stacking and perfusing of living building parts for solid organ fabrication.
According to the authors, a “grand challenge” of tissue engineering has been to construct large living structures with high cell density akin to native organs and to sustain the viability of the structures while reproducing in vivo function. Scaffold-based tissue engineering methods usually involve either decellularizing a native organ and reseeding the residual extracellular matrix with the desired cells or seeding a biocompatible fabricated scaffold with cells. Scaffold-free methods, including pellet culture and hanging drops, are generally limited by the small scale of the produced tissue construct. Cell sheets, while larger in scale, have had only limited success in specific applications such as vascular tissue engineering.
The potential clinical impact of engineered tissues in treating disease is vast, the paper read. Liver failure, for example, leads to 1 million deaths per year worldwide, the 10th most common overall cause of death. This represents enormous direct and indirect healthcare costs. The supply of donor organs is far too small to meet the need, and many patients die while waiting on the transplant list. As the obesity epidemic increases, nonalcoholic fatty liver disease leading to liver failure will become an even more important clinical issue. So manufacturing live tissue by any means would count as a major breakthrough for a swath of applications.
The Bio-P3 builds on the principles of the pick and place devices used in the high-speed assembly of multicomponent electronics. The authors position the device as having exciting potential applications in solid organ tissue engineering as a platform for applications such as organ function replacement, solid tumor modeling, or three-dimensional drug testing.
“This device can be the long-expected breakthrough in the field of regenerative medicine and hopefully allow the fabrication of large 3-D organs and tissues," says John A. Jansen, DDS, Ph.D., co-editor-in-chief of Tissue Engineering, Part C: Methods and professor and head of dentistry, Radboud University Medical Center, The Netherlands.