01.26.15
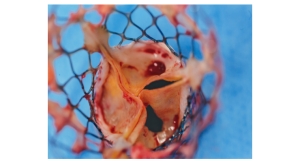
Italian cardiovascular device company Sorin Group reported the completion of enrollment for the Perceval investigation device exemption (IDE) Trial. The trial enrollment was completed on schedule with the implantation of 300 patients.
The IDE study, which was completed with the implantation of 300 patients, was designed as a prospective, non-randomized, multi-center clinical trial. The purpose of the study is to demonstrate the safety and effectiveness of the Perceval valve to historic controls with the device when used to replace diseased or dysfunctional aortic valves or aortic valve prostheses. The patients enrolled in the U.S. IDE study were all adult patients suffering from aortic valve stenosis or steno-insufficiency, indicated for native or prosthetic aortic valve replacement and available for postoperative follow-up beyond one year. In total, 18 centers across the United States took part in the study with the first patient enrolled in June 2013.
The principal U.S. investigator of the study is Rakesh Suri, M.D., Ph.D., with the Mayo Clinic in Rochester, Minnesota.
“We are very excited to have completed this important milestone which wouldn’t have been possible without the outstanding involvement of this study’s investigators under the leadership of Dr Suri. We now very much look forward to the results,” said Michel Darnaud, Sorin Group, president, Cardiac Surgery Business Unit. “The Perceval valve once approved in the U.S. will complement Sorin’s already strong portfolio of pericardial aortic valve solutions, including the Solo Smart and Mitroflow PRT valves.”
For the patients who remain candidates for traditional surgical aortic valve replacement, the Perceval valve offers the potential benefit of shortening the time spent supported by the heart-lung machine. All this is performed without the need for sutures around the valve, thereby offering the possibility of shorter operative times, decreased complication rates, and use of minimally invasive approaches.
Results from a similar study conducted in Europe were presented at the recent European Association for Cardio-Thoracic Surgery annual meeting in Milan, Italy. The Perceval Cavalier trial included results from a pooled European multi-center experience. The trial was designed to evaluate the safety and effectiveness of the Perceval valve in a large population requiring aortic valve replacement with or without concomitant coronary bypass grafting. A total of 658 patients were implanted at 25 European centers. The 30-day results, presented by Francois Laborde, M.D., professor of cardiac surgery at the Institut Mutualiste Montsouris in Paris, France, demonstrated the safety of Perceval with “excellent hemodynamics, a low complication rate, and ease of implant with a reproducible technique in both traditional sternotomy and the less-invasive approach,” according to the company.
In a second study—a European multi-center experience with suture-less Perceval valve, measuring clinical and haemodynamic outcomes up to five years in more than 700 patients—Perceval demonstrated “excellent and stable results” at up to five years of follow-up, Sorin officials reported. According to the company, there was no valve migration, structural valve degeneration or valve thrombosis was observed in the largest patient cohort ever implanted with sutureless valves. Low early 30-day mortality (1.9 percent), low major early paravalvular leak (1.4 percent) and low early stroke (1.6 percent) suggested favorable safety and efficacy profile of the Perceval valve. An additional benefit was the very low endocarditis rate of 0.3 percent.
According to the company, a recent study in Belgium evaluated the economic impact of the Perceval sutureless valves compared to traditional stented valves (Perimount valve from Edwards Lifescience). The mean total cost of the Perceval patients group compared with Perimount, net of valve costs, showed a reduction of 27 percent, i.e., a mean reduction of $9,635 per case. The cost savings mainly were driven by reduced hospital stay costs and significantly lower procedural costs, the company reported.
In line with the European findings, a recently published article1 reported the first successful experiences with Perceval in the United States, highlighting how the absence of major surgical complications, identifiable reductions in bypass/cross-clamp times, and complete absence of paravalvular leak are potentially appealing features of this novel device for severe calcific aortic valve stenosis.1
Perceval now has been implanted in more than 8,000 patients in more tham 250 centers, in 44 countries.
Sorin Group focuses on two major therapeutic areas: Cardiac Surgery (cardiopulmonary products for open heart surgery and heart valve repair or replacement prostheses) and Cardiac Rhythm Management (pacemakers, defibrillators and non invasive monitoring to diagnose arrhythmias and deliver anti-arrhythmia therapies as well as cardiac resynchronization devices for heart failure treatment).
Reference:
1. Hector I. Michelena et al, An Alternative for Surgical Management of Calcific Aortic Valve Stenosis: Sutureless Valve Implants, J Card Surg 2014;XX
The IDE study, which was completed with the implantation of 300 patients, was designed as a prospective, non-randomized, multi-center clinical trial. The purpose of the study is to demonstrate the safety and effectiveness of the Perceval valve to historic controls with the device when used to replace diseased or dysfunctional aortic valves or aortic valve prostheses. The patients enrolled in the U.S. IDE study were all adult patients suffering from aortic valve stenosis or steno-insufficiency, indicated for native or prosthetic aortic valve replacement and available for postoperative follow-up beyond one year. In total, 18 centers across the United States took part in the study with the first patient enrolled in June 2013.
The principal U.S. investigator of the study is Rakesh Suri, M.D., Ph.D., with the Mayo Clinic in Rochester, Minnesota.
“We are very excited to have completed this important milestone which wouldn’t have been possible without the outstanding involvement of this study’s investigators under the leadership of Dr Suri. We now very much look forward to the results,” said Michel Darnaud, Sorin Group, president, Cardiac Surgery Business Unit. “The Perceval valve once approved in the U.S. will complement Sorin’s already strong portfolio of pericardial aortic valve solutions, including the Solo Smart and Mitroflow PRT valves.”
For the patients who remain candidates for traditional surgical aortic valve replacement, the Perceval valve offers the potential benefit of shortening the time spent supported by the heart-lung machine. All this is performed without the need for sutures around the valve, thereby offering the possibility of shorter operative times, decreased complication rates, and use of minimally invasive approaches.
Results from a similar study conducted in Europe were presented at the recent European Association for Cardio-Thoracic Surgery annual meeting in Milan, Italy. The Perceval Cavalier trial included results from a pooled European multi-center experience. The trial was designed to evaluate the safety and effectiveness of the Perceval valve in a large population requiring aortic valve replacement with or without concomitant coronary bypass grafting. A total of 658 patients were implanted at 25 European centers. The 30-day results, presented by Francois Laborde, M.D., professor of cardiac surgery at the Institut Mutualiste Montsouris in Paris, France, demonstrated the safety of Perceval with “excellent hemodynamics, a low complication rate, and ease of implant with a reproducible technique in both traditional sternotomy and the less-invasive approach,” according to the company.
In a second study—a European multi-center experience with suture-less Perceval valve, measuring clinical and haemodynamic outcomes up to five years in more than 700 patients—Perceval demonstrated “excellent and stable results” at up to five years of follow-up, Sorin officials reported. According to the company, there was no valve migration, structural valve degeneration or valve thrombosis was observed in the largest patient cohort ever implanted with sutureless valves. Low early 30-day mortality (1.9 percent), low major early paravalvular leak (1.4 percent) and low early stroke (1.6 percent) suggested favorable safety and efficacy profile of the Perceval valve. An additional benefit was the very low endocarditis rate of 0.3 percent.
According to the company, a recent study in Belgium evaluated the economic impact of the Perceval sutureless valves compared to traditional stented valves (Perimount valve from Edwards Lifescience). The mean total cost of the Perceval patients group compared with Perimount, net of valve costs, showed a reduction of 27 percent, i.e., a mean reduction of $9,635 per case. The cost savings mainly were driven by reduced hospital stay costs and significantly lower procedural costs, the company reported.
In line with the European findings, a recently published article1 reported the first successful experiences with Perceval in the United States, highlighting how the absence of major surgical complications, identifiable reductions in bypass/cross-clamp times, and complete absence of paravalvular leak are potentially appealing features of this novel device for severe calcific aortic valve stenosis.1
Perceval now has been implanted in more than 8,000 patients in more tham 250 centers, in 44 countries.
Sorin Group focuses on two major therapeutic areas: Cardiac Surgery (cardiopulmonary products for open heart surgery and heart valve repair or replacement prostheses) and Cardiac Rhythm Management (pacemakers, defibrillators and non invasive monitoring to diagnose arrhythmias and deliver anti-arrhythmia therapies as well as cardiac resynchronization devices for heart failure treatment).
Reference:
1. Hector I. Michelena et al, An Alternative for Surgical Management of Calcific Aortic Valve Stenosis: Sutureless Valve Implants, J Card Surg 2014;XX