11.20.14
St. Jude Medical Inc. reported retrospective data analysis from the Champion clinical trial showing significant a reduction in 30-day hospital readmission rates for patients age 65 and older.
The trial looked at the safety and effectiveness of the CardioMEMS HF System for patients with New York Heart Association (NYHA) Class III heart failure (HF) who had been hospitalized for HF in the previous 12 months. The analysis, titled "Impact of Wireless Pulmonary Artery Pressure Monitoring on Heart Failure Hospitalization and All-Cause 30-Day Readmissions in Medicare-Eligible Patients with NYHA Class III Heart Failure," was presented during the American Heart Association Scientific Sessions.
This analysis looked at patients 65 and older (Medicare-eligible) from the Champion trial and found that there was a 58 percent reduction in all-cause hospital readmissions (hospitalization for any reason) and a 78 percent reduction in HF hospital readmissions when patients were managed with the CardioMEMS HF System compared to standard-of-care medical management.
The suggest that use of the CardioMEMS HF System can significantly reduce the hospital’s risk of government-imposed penalties that are designed to reduce patient readmissions within 30 days of treatment.
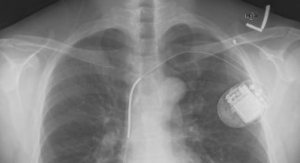
The CardioMEMS system uses a miniaturized, wireless monitoring sensor that is implanted in the pulmonary artery (PA) during a minimally invasive procedure to directly measure PA pressure. Measuring PA pressure allows clinicians to proactively manage treatment with medication changes for patients with worsening HF before visible symptoms, such as weight and blood pressure changes, occur. The system allows patients to transmit PA pressure data from their homes to their healthcare providers, who then manage appropriate medication changes to reduce the likelihood of hospitalization.
Under the U.S. Centers for Medicare and Medicaid Services (CMS) Hospital Readmissions Reduction Program, hospitals with excess all-cause 30-day readmissions (a hospital-specific CMS adjustment compared with the national average) for HF, heart attack and pneumonia discharges were penalized $227 million for fiscal year 2014. The penalty fees list in 2015 also will include chronic obstructive pulmonary disease, total hip and total knee replacement and the estimated penalties are projected at $428 million. The hospital penalties extend across total Medicare payments based on the performance of these specific conditions.
“Incorporating proactive pulmonary artery pressure monitoring into patient management significantly reduced heart failure hospitalizations and 30-day all-cause and HF readmissions in patients age 65 and older. The adoption of this treatment strategy using the CardioMEMS HF System addresses the unmet need within the U.S. healthcare system for hospitals struggling to meet the requirements of CMS,” said Philip Adamson, M.D., MC director of the Heart Failure Institute at Oklahoma Heart Hospital in Oklahoma City.
“These data are recognition of the importance the CardioMEMS HF System brings to heart failure patients burdened with multiple hospital admissions each year and the cost savings it brings to the healthcare system,” said Mark D. Carlson, M.D., chief medical officer for St. Jude Medical. “We continue our commitment of investing in meaningful clinical research with this new data analysis from the Champion trial that is helping shape the way physicians are caring for their patients.”
Heart failure occurs when the heart is unable to pump enough blood to meet the body’s demands and blood pressure within the heart is elevated. Significant HF progression over a period of days is known as acute decompensation and leads to hospitalization. Increased PA pressures often precede indirect measures of worsening HF such as weight and blood pressure changes.
The CardioMEMS HF System, approved by the U.S. Food and Drug Administration (FDA) in May, allows clinicians to stabilize PA pressures by proactively managing medications and other treatment options while also providing an early indication of worsening HF.
St. Jude Medical purchased the remaining shares of CardioMEMS in May—soon after FDA approval of the company’s primary technology—for $375 million. In September 2010, St. Jude paid $60 million for a 19 percent stake in CardioMEMS, with an exclusive option to buy the remaining 81 percent.
Approximately 26 million people worldwide suffer from heart failure.
Headquartered in St. Paul, Minn., St. Jude Medical has four major clinical focus areas that include cardiac rhythm management, atrial fibrillation, cardiovascular and neuromodulation.
The trial looked at the safety and effectiveness of the CardioMEMS HF System for patients with New York Heart Association (NYHA) Class III heart failure (HF) who had been hospitalized for HF in the previous 12 months. The analysis, titled "Impact of Wireless Pulmonary Artery Pressure Monitoring on Heart Failure Hospitalization and All-Cause 30-Day Readmissions in Medicare-Eligible Patients with NYHA Class III Heart Failure," was presented during the American Heart Association Scientific Sessions.
This analysis looked at patients 65 and older (Medicare-eligible) from the Champion trial and found that there was a 58 percent reduction in all-cause hospital readmissions (hospitalization for any reason) and a 78 percent reduction in HF hospital readmissions when patients were managed with the CardioMEMS HF System compared to standard-of-care medical management.
The suggest that use of the CardioMEMS HF System can significantly reduce the hospital’s risk of government-imposed penalties that are designed to reduce patient readmissions within 30 days of treatment.
The CardioMEMS system uses a miniaturized, wireless monitoring sensor that is implanted in the pulmonary artery (PA) during a minimally invasive procedure to directly measure PA pressure. Measuring PA pressure allows clinicians to proactively manage treatment with medication changes for patients with worsening HF before visible symptoms, such as weight and blood pressure changes, occur. The system allows patients to transmit PA pressure data from their homes to their healthcare providers, who then manage appropriate medication changes to reduce the likelihood of hospitalization.
Under the U.S. Centers for Medicare and Medicaid Services (CMS) Hospital Readmissions Reduction Program, hospitals with excess all-cause 30-day readmissions (a hospital-specific CMS adjustment compared with the national average) for HF, heart attack and pneumonia discharges were penalized $227 million for fiscal year 2014. The penalty fees list in 2015 also will include chronic obstructive pulmonary disease, total hip and total knee replacement and the estimated penalties are projected at $428 million. The hospital penalties extend across total Medicare payments based on the performance of these specific conditions.
“Incorporating proactive pulmonary artery pressure monitoring into patient management significantly reduced heart failure hospitalizations and 30-day all-cause and HF readmissions in patients age 65 and older. The adoption of this treatment strategy using the CardioMEMS HF System addresses the unmet need within the U.S. healthcare system for hospitals struggling to meet the requirements of CMS,” said Philip Adamson, M.D., MC director of the Heart Failure Institute at Oklahoma Heart Hospital in Oklahoma City.
“These data are recognition of the importance the CardioMEMS HF System brings to heart failure patients burdened with multiple hospital admissions each year and the cost savings it brings to the healthcare system,” said Mark D. Carlson, M.D., chief medical officer for St. Jude Medical. “We continue our commitment of investing in meaningful clinical research with this new data analysis from the Champion trial that is helping shape the way physicians are caring for their patients.”
Heart failure occurs when the heart is unable to pump enough blood to meet the body’s demands and blood pressure within the heart is elevated. Significant HF progression over a period of days is known as acute decompensation and leads to hospitalization. Increased PA pressures often precede indirect measures of worsening HF such as weight and blood pressure changes.
The CardioMEMS HF System, approved by the U.S. Food and Drug Administration (FDA) in May, allows clinicians to stabilize PA pressures by proactively managing medications and other treatment options while also providing an early indication of worsening HF.
St. Jude Medical purchased the remaining shares of CardioMEMS in May—soon after FDA approval of the company’s primary technology—for $375 million. In September 2010, St. Jude paid $60 million for a 19 percent stake in CardioMEMS, with an exclusive option to buy the remaining 81 percent.
Approximately 26 million people worldwide suffer from heart failure.
Headquartered in St. Paul, Minn., St. Jude Medical has four major clinical focus areas that include cardiac rhythm management, atrial fibrillation, cardiovascular and neuromodulation.