There are only a few big medical device companies that dominate the continuous glucose monitoring space and Senseonics is hoping to join the ranks. The firm has raised $20 million from current investors to advance an implant that continuously monitors blood-glucose levels and transmits the data to patients and doctors through a smartphone app.
Although the Germantown, Md.-based company has existed for 17 years, it has spent most of that time focused on research. Only in the past few years with the hire of CEO Tim Goodnow has the company focused its attention on advancing medical devices. Goodnow previously worked at Abbott Diabetes Care where he was vice president for technical operations.
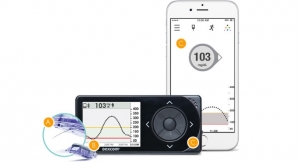
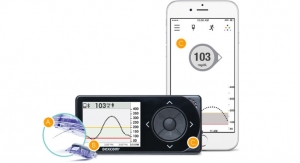
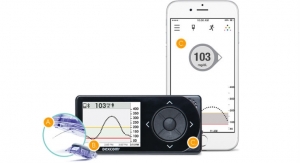
Senseonics sensor is implanted in the upper arm for up to six months. It uses a fluorescent, glucose-indicating polymer. A light emitting diode in the sensor stimulates the polymer, and the polymer signals changes in glucose concentration through a change in the light. The glucose measurement is relayed to a transmitter. This entire measurement is designed to be done autonomously and independently, according to the description of the product on the company’s website.
Pivotal trials have begun in Amsterdam and should take about one year. After it secures a greenlight for its investigational device exemption application, Senseonics expects to begin clinical trials in the United States next year. So far, 200 patients have tried the device.
Among Sensionics’ investors are Abingworth Management, Anthem Capital, Delphi Ventures, Greenspring Associates, Healthcare Ventures, New Enterprise Associates and Rho Capital Partners.
The company is aiming for more accurate readings than those produced by Dexcom, Medtronic Inc. and Abbott Laboratories, the latter of which has a device in the European market. Reportlinker projects the continuous glucose monitoring device segment to reach $568.5 million by 2020.
In an interview with MedCity News, Mirasol Panlilio, Senseonics vice president of marketing and sales, said the device goes beyond the current standard for continuous glucose monitoring devices. It leaves out the handheld device that current models tend to use. The sensor talks to the transmitter and does the calculations and the data is sent to either an iPhone or Android.
Panlilio said the target patient population is Type 1 and Type 2 diabetes patients who want to better manage their condition.