04.16.14
The U.S. Patent and Trademark Office has issued a new patent to Reverse Medical Corporation for its Micro Vascular Plug (MVP) technology.
The new patent, “Embolic Implant and Method of Use,” allows 22 claims surrounding the company’s MVP IP portfolio.
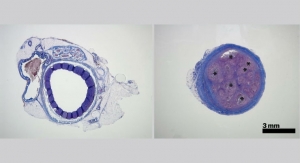
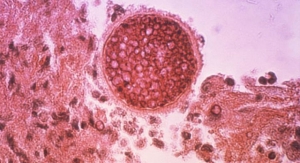
The Reverse Medical MVP represents a proprietary technology designed specifically for vascular embolization. A number of clinical applications require occlusion of the vasculature to rapidly, effectively and safely provide blood flow cessation, including presentations of trauma, hemorrhage, tumor and vascular malformation isolations.
Physicians traditionally have used embolic coils for vascular embolization, often requiring numerous costly implants, extensive endovascular lab time and considerable fluoroscopic radiation exposure to both the patient and the treating medical staff. The Reverse Medical MVP rapidly occludes target blood vessels, most often with only one single implant. Furthermore, the MVP is uniquely deliverable through traditional microcatheters, allowing the physician to super-selectively perform rapid vascular embolization at the target vessel location. The MVP technology platform will be offered in a variety of sizes to treat all areas of the body’s peripheral vasculature, according to Reverse Medical.
The newly-issued patent builds upon Reverse’s intellectual property portfolio, which now includes six issued patents and 13 additional pending patent applications worldwide.
“This patent solidifies our intellectual property foundation of unique means for performing endovascular embolization and, along with our other issued patents and pending applications, provides us with broad protection for our innovative portfolio of devices for treating a variety of Neuro and Peripheral vascular conditions,” Reverse Medical Chief Technology Officer Brian Strauss said.
Reverse Medical Corporation is a privately held medical device company that develops endovascular treatments for various peripheral and neurovascular disorders and diseases. The firm is based in Irvine, Calif.
The new patent, “Embolic Implant and Method of Use,” allows 22 claims surrounding the company’s MVP IP portfolio.
The Reverse Medical MVP represents a proprietary technology designed specifically for vascular embolization. A number of clinical applications require occlusion of the vasculature to rapidly, effectively and safely provide blood flow cessation, including presentations of trauma, hemorrhage, tumor and vascular malformation isolations.
Physicians traditionally have used embolic coils for vascular embolization, often requiring numerous costly implants, extensive endovascular lab time and considerable fluoroscopic radiation exposure to both the patient and the treating medical staff. The Reverse Medical MVP rapidly occludes target blood vessels, most often with only one single implant. Furthermore, the MVP is uniquely deliverable through traditional microcatheters, allowing the physician to super-selectively perform rapid vascular embolization at the target vessel location. The MVP technology platform will be offered in a variety of sizes to treat all areas of the body’s peripheral vasculature, according to Reverse Medical.
The newly-issued patent builds upon Reverse’s intellectual property portfolio, which now includes six issued patents and 13 additional pending patent applications worldwide.
“This patent solidifies our intellectual property foundation of unique means for performing endovascular embolization and, along with our other issued patents and pending applications, provides us with broad protection for our innovative portfolio of devices for treating a variety of Neuro and Peripheral vascular conditions,” Reverse Medical Chief Technology Officer Brian Strauss said.
Reverse Medical Corporation is a privately held medical device company that develops endovascular treatments for various peripheral and neurovascular disorders and diseases. The firm is based in Irvine, Calif.
that develops endovascular treatments for various peripheral and neurovascular disorders and diseases. The company is based in Irvine, California. - See more at: http://www.mpo-mag.com/contents/view_breaking-news/2014-02-04/reverse-medical-receives-fda-clearance-of-micro-vascular-plug/#sthash.DGkhaELo.dpuf