Niki Arrowsmith05.01.13
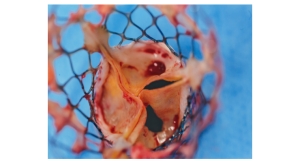
The U.S. Food and Drug Administration has accepted and approved Sorin Group’s Investigational Device Exemption (IDE) application and clinical trial protocol to begin evaluating its Perceval S suture-less aortic tissue valve (pictured left).
Rakesh Suri, M.D., associate professor of surgery and consultant cardiovascular surgeon at the Mayo Clinic, Rochester, Minn., is principal investigator for the Perceval S IDE trial, which will involve up to 25 U.S. centers. The purpose of this prospective, non-randomized, multi-center clinical trial is to demonstrate the safety and effectiveness of the Perceval S suture-less heart valve when used to replace a diseased or dysfunctional aortic valve or aortic valve prosthesis.
Perceval S is a bioprosthetic valve designed to replace a diseased native or malfunctioning prosthetic aortic valve in patients with aortic stenosis using either traditional or minimally invasive heart surgery. Due to its original characteristic of allowing suture-less positioning and anchoring at the implantation site, the Perceval S aortic valve will offer significant advancements in surgical aortic valve replacement (AVR) technology.
Aortic stenosis, one of the most common valvular heart abnormalities, is a degenerative disease resulting from a progressive age-dependent build-up of calcium that disrupts blood flow across the aortic valve.
Perceval S is designed to be implanted through a traditional open-heart surgical approach, through a minimally invasive cardiac surgery (MICS) partial sternotomy (where a fine incision is made into the sternum) or using a right mini-thoracotomy implantation technique (an incision into the pleural cavity of the chest). MICS techniques are increasingly important for AVR because they reduce surgical trauma and morbidity, an important consideration for the growing number of AVR patients.
Perceval S earned the CE mark for sale in the European Union in 2011, and had been implanted more than 2,000 times, according to the company.
“We are eager to provide this innovative treatment to a wider patient population in the United States and to support the cardiac surgeon community in performing more and more MICS procedures,” said Michel Darnaud, president of the cardiac surgery business unit at Sorin Group.
Sorin Group’s U.S. headquarters are located in Arvada, Colo. The company is based in Milan, Italy.
Rakesh Suri, M.D., associate professor of surgery and consultant cardiovascular surgeon at the Mayo Clinic, Rochester, Minn., is principal investigator for the Perceval S IDE trial, which will involve up to 25 U.S. centers. The purpose of this prospective, non-randomized, multi-center clinical trial is to demonstrate the safety and effectiveness of the Perceval S suture-less heart valve when used to replace a diseased or dysfunctional aortic valve or aortic valve prosthesis.
Perceval S is a bioprosthetic valve designed to replace a diseased native or malfunctioning prosthetic aortic valve in patients with aortic stenosis using either traditional or minimally invasive heart surgery. Due to its original characteristic of allowing suture-less positioning and anchoring at the implantation site, the Perceval S aortic valve will offer significant advancements in surgical aortic valve replacement (AVR) technology.
Aortic stenosis, one of the most common valvular heart abnormalities, is a degenerative disease resulting from a progressive age-dependent build-up of calcium that disrupts blood flow across the aortic valve.
Perceval S is designed to be implanted through a traditional open-heart surgical approach, through a minimally invasive cardiac surgery (MICS) partial sternotomy (where a fine incision is made into the sternum) or using a right mini-thoracotomy implantation technique (an incision into the pleural cavity of the chest). MICS techniques are increasingly important for AVR because they reduce surgical trauma and morbidity, an important consideration for the growing number of AVR patients.
Perceval S earned the CE mark for sale in the European Union in 2011, and had been implanted more than 2,000 times, according to the company.
“We are eager to provide this innovative treatment to a wider patient population in the United States and to support the cardiac surgeon community in performing more and more MICS procedures,” said Michel Darnaud, president of the cardiac surgery business unit at Sorin Group.
Sorin Group’s U.S. headquarters are located in Arvada, Colo. The company is based in Milan, Italy.